Thermal vs. Non-Thermal Food Processing: Impact on Bioactive Compounds for Biomedical Applications
This article provides a comprehensive analysis of how thermal and non-thermal food processing technologies influence the stability, bioavailability, and bioactivity of phytochemicals.
Thermal vs. Non-Thermal Food Processing: Impact on Bioactive Compounds for Biomedical Applications
Abstract
This article provides a comprehensive analysis of how thermal and non-thermal food processing technologies influence the stability, bioavailability, and bioactivity of phytochemicals. Tailored for researchers, scientists, and drug development professionals, it explores the fundamental mechanisms of nutrient degradation and preservation, details the operation and food applications of emerging non-thermal methods, and addresses key challenges in scaling these technologies. A critical comparative synthesis evaluates the evidence on efficacy for retaining bioactivity, offering validated insights to guide the selection of processing methods for functional food development and nutraceutical extraction.
The Fundamental Divide: How Heat and Non-Thermal Energy Affect Bioactive Compounds
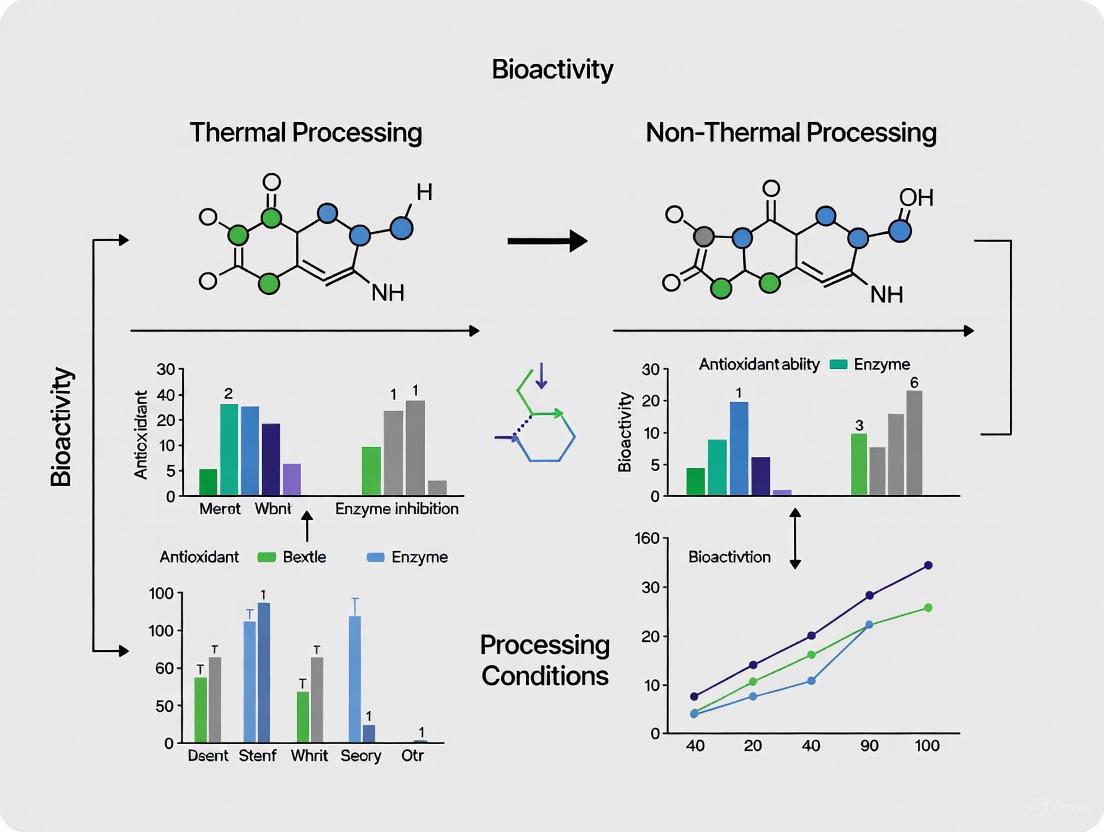
Thermal processing remains a cornerstone of food manufacturing, serving as a critical intervention to ensure microbial safety and extend product shelf-life. However, this essential safety measure introduces a significant paradox: the same heat that inactivates pathogens also degrades heat-sensitive nutrients and bioactive compounds. The thermal degradation of vitamins, antioxidants, and unsaturated lipids presents a fundamental challenge to nutritional quality [1]. Conversely, emerging non-thermal technologies propose an alternative pathway, aiming to achieve comparable safety standards while better preserving the nutritional integrity of food products [2]. This comparative analysis objectively examines the scientific evidence underlying both approaches, providing researchers and product developers with quantitative data on their respective impacts on bioactivity and safety parameters.
Comparative Mechanisms of Action
Fundamental Principles of Thermal Processing
Conventional thermal processing, including pasteurization and sterilization, relies on the transfer of thermal energy to food matrices. The efficacy of microbial destruction and nutrient degradation is governed by time-temperature relationships, quantified through the thermal processing level (F-value) and cook value (C-value) [3]. The underlying principle of High-Temperature Short-Time processing leverages the differential between the z-values of microorganisms (typically 7-12°C) and nutrients (typically 25-45°C) [3]. This differential suggests that higher temperatures applied for shorter durations can achieve equivalent microbial lethality while reducing nutritional damage. However, thermal processing inevitably initiates Maillard reactions, lipid oxidation, and protein denaturation, leading to both desirable sensory attributes and undesirable nutrient loss or contaminant formation [4].
Fundamental Principles of Non-Thermal Processing
Non-thermal technologies achieve microbial safety through physical or chemical mechanisms that do not primarily rely on heat. These methods operate at or near ambient temperatures, thereby minimizing thermal degradation pathways. Key technologies include:
- High Hydrostatic Pressure: Applies isostatic pressure (100-600 MPa) to inactivate microorganisms through protein denaturation and cell membrane damage while preserving small molecules like vitamins and pigments [2].
- Pulsed Electric Fields: Utilizes short, high-voltage pulses to induce electroporation of microbial cell membranes, leading to cell death while minimally affecting nutritional compounds [5].
- Cold Plasma: Generates reactive oxygen and nitrogen species through ionized gases that oxidize microbial components without significantly elevating product temperature [2].
- Ultrasonication: Employs high-frequency sound waves to create cavitation bubbles whose collapse generates localized energy that disrupts microbial cells and enhances extraction of bioactives [5].
Table 1: Fundamental Mechanisms of Non-Thermal Processing Technologies
| Technology | Primary Inactivation Mechanism | Typical Operating Parameters | Key Physical Effects |
|---|---|---|---|
| High Hydrostatic Pressure | Protein denaturation, cell membrane damage | 100-600 MPa, ambient to 60°C | Isostatic pressure transmission, minimal effect on covalent bonds |
| Pulsed Electric Fields | Electroporation of cell membranes | 10-80 kV/cm, short pulses (µs-ms) | Electrical field-induced pore formation |
| Cold Plasma | Oxidative damage by reactive species | Various plasma sources (DBD, jet) | Generation of ROS/RNS, UV photons, charged particles |
| Ultrasonication | Cavitation-induced cell disruption | 20-100 kHz, variable amplitude | Microjet formation, shear forces, localized heating |
Impact on Nutritional Components: Comparative Data
Vitamin and Phytochemical Stability
Thermal processing significantly degrades heat-labile nutrients, with water-soluble vitamins being particularly vulnerable. Research demonstrates that vitamin C losses range from 10% to over 90% depending on processing intensity, while thiamine (B1) shows extreme sensitivity in neutral and alkaline environments [1]. Similarly, phytochemicals like (−)-epicatechin experience rapid degradation during thermal treatment, with studies showing approximately 65.2% loss within the first 10 minutes of heating in aqueous solution, and over 99.5% degradation within 30 minutes [6].
In contrast, non-thermal treatments generally demonstrate superior retention of these compounds. For instance, thermo-sonication combined with nisin in orange juice increased total polyphenols by 10.03% and total carotenoids by 20.10%, while largely retaining ascorbic acid and flavonoids [7]. High-pressure processing of potatoes showed no changes in total phenolic content or antioxidant activity, although specific phenolic acids demonstrated varying sensitivity [7].
Protein and Bioactive Compound Integrity
The structural and functional properties of proteins are notably affected by excessive heat treatment. Studies on donkey milk whey proteins revealed that high-temperature long-time treatment resulted in decreased fluorescence intensity, foaming stability, and emulsification stability, with considerable damage to active protein components [8]. Specifically, lysozyme activity preservation was only 23%, while lactoferrin and IgG exhibited significant losses of 70% and 77%, respectively [8].
Non-thermal treatment methods showed superior efficacy in preserving these bioactive components. Ultrasonic treatment diminished protein particle size and turbidity, while UV treatment oxidized internal disulfide bonds within proteins, consequently augmenting free sulfhydryl groups beneficial to foaming and emulsification stability [8].
Table 2: Comparative Impact on Bioactive Compounds: Thermal vs. Non-Thermal Processing
| Bioactive Compound | Thermal Processing Impact | Non-Thermal Processing Impact | Experimental Context |
|---|---|---|---|
| Vitamin C | 10-90% loss | Largely retained | Orange juice processing [7] [1] |
| (−)-Epicatechin | 65.2% loss in 10 min | Not quantified | Aqueous solution at boiling temperature [6] |
| Total Polyphenols | Variable degradation | 10.03% increase | Orange juice with thermo-sonication + nisin [7] |
| Total Carotenoids | Thermal isomerization | 20.10% increase | Orange juice with thermo-sonication + nisin [7] |
| Lysozyme Activity | 77% loss | Significantly preserved | Donkey milk whey proteins [8] |
| Lactoferrin | 70% loss | Significantly preserved | Donkey milk whey proteins [8] |
| Polyunsaturated Fatty Acids | Serious thermal damage, oxidation | Reduced oxidation | Pacific saury fillets [3] |
Fatty Acid Stability
Fatty acids, particularly polyunsaturated fatty acids (PUFAs) like EPA and DHA, are highly susceptible to oxidation during thermal processing. Research on Pacific saury fillets demonstrated that higher maximum processing temperatures caused serious thermal damage to fatty acids, especially PUFAs [3]. The extraction coefficient of lipids and fatty acids decreased with increasing heating rates, indicating less thermal damage to fish tissue at faster heating rates [3].
HTST processing with extra high temperature brought significant damage to fatty acids, though higher heating rates associated with technologies like microwave heating could potentially reduce this damage [3]. The formation of trans fatty acids during thermal processing further compounds the nutritional concerns, with epidemiological studies linking them to increased breast cancer risk [4].
Microbial Safety and Contaminant Formation
Pathogen Inactivation Efficacy
Thermal processing reliably achieves microbial safety through well-characterized time-temperature relationships. The thermal processing level (F-value) calculates cumulative thermal lethality based on the time-temperature profile at the cold spot, typically targeting a minimum of F₀ = 3.0 minutes for low-acid canned foods, with commercial operations often applying F₀ = 6.0 minutes or longer for an additional safety margin [3].
Non-thermal technologies achieve microbial inactivation through different mechanisms, with varying efficacy depending on the technology and processing parameters. High Hydrostatic Pressure (100-600 MPa) effectively inactivates vegetative microorganisms through protein denaturation and cell membrane damage [2]. Pulsed Electric Fields (10-80 kV/cm) induce electroporation of bacterial cell membranes [5]. Cold Plasma generates reactive oxygen and nitrogen species that oxidize microbial components [2]. Ultrasonication creates cavitation that physically disrupts microbial cells [5].
Processing-Induced Contaminants
Thermal processing can generate various hazardous compounds via Maillard reactions, lipid oxidation, and thermal degradation. These include polycyclic aromatic hydrocarbons, heterocyclic aromatic amines, acrylamide, furan, trans fatty acids, and advanced glycation end-products [4]. The International Agency for Research on Cancer classifies several of these compounds as known or probable carcinogens, with epidemiological studies linking them to increased cancer incidence [4].
Non-thermal technologies generally produce fewer or different processing contaminants. However, some non-thermal methods may induce oxidative changes; for instance, medium-frequency ultrasonication can generate radical species that cause oxidative changes in lipids and proteins [5]. Cold plasma's reactive species may also potentially modify food components, though typically to a lesser extent than thermal treatment.
Experimental Protocols and Methodologies
Thermal Processing Experimental Framework
Protocol 1: High-Temperature Short-Time (HTST) Processing of Solid Foods
- Sample Preparation: Pacific saury fillets are cut to standardized dimensions (40×30×6 mm) and vacuum-packaged in retortable pouches with mobile metallic temperature sensors inserted at the cold spot [3].
- Processing System: A specialized thermal processing system consisting of an oil bath heat source and pressure-proof container is used. The container is placed in preheated oil to achieve rapid heat transfer [3].
- Parameter Control: Heating rates (5.48-18.30°C/min), maximum temperatures (123°C, 133°C), and thermal processing levels (F₀ = 3.0 min, 6.0 min) are carefully controlled and monitored [3].
- Analysis: Fatty acid composition is analyzed via chromatography, with extraction coefficients calculated to quantify thermal damage [3].
Protocol 2: Thermal Stability Assessment of Phytochemicals
- Experimental Setup: (−)-Epicatechin solutions are subjected to boiling water bath conditions for up to 85 minutes to simulate thermal processing conditions [6].
- Monitoring: Degradation products are analyzed using UPLC-ESI-TSQ-MS/MS at multiple time points to determine degradation kinetics [6].
- Product Identification: Degradation products are identified based on retention time, full scan, and tandem MS data, with isomerization, oxidation, hydroxylation, dimerization, and ring cleavage reactions characterized [6].
Non-Thermal Processing Experimental Framework
Protocol 3: High-Pressure Processing of Fruit/Vegetable Products
- Sample Preparation: Fresh produce is washed, sliced if necessary, and vacuum-sealed in flexible packaging compatible with high-pressure processing [7].
- Processing Parameters: Pressure levels (400-600 MPa), holding time (3-5 minutes), and temperature (ambient or controlled) are applied using industrial HPP equipment [7] [2].
- Quality Assessment: Polyphenol oxidase activity, total phenolic content, antioxidant activity, and individual phenolic compounds are quantified before and after processing [7].
- Storage Studies: Processed products are stored under controlled conditions with periodic analysis of bioactive compounds and microbial quality [7].
Protocol 4: Pulsed Electric Field Treatment of Liquid Foods
- System Configuration: A PEF system with parallel electrode treatment chamber, high-voltage pulse generator, and temperature control system is used [5].
- Processing Parameters: Electric field strength (3.5 kV/cm), pulse number (e.g., 5 pulses), and specific energy input (0.6 kJ/kg) are optimized for different matrices [7].
- Bioaccessibility Assessment: Treated samples are subjected to in vitro digestion models to determine bioactive compound bioaccessibility [7].
- Comparative Analysis: PEF-treated samples are compared with thermally pasteurized counterparts for nutrient retention and microbial safety [7].
Visualization of Processing Pathways and Effects
Thermal Processing Impact Pathways
Thermal Processing Dual Effects
Non-Thermal Processing Mechanisms
Non-Thermal Processing Benefits
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Research Reagents and Materials for Processing Studies
| Reagent/Material | Function in Research | Application Examples |
|---|---|---|
| UPLC-ESI-TSQ-MS/MS | Identification and quantification of degradation products | Monitoring (−)-epicatechin degradation products [6] |
| HPLC with UV/Fluorescence Detection | Quantification of water-soluble and fat-soluble vitamins | Analysis of vitamin retention in processed foods [1] |
| Mobile Metallic Temperature Sensors | Accurate time-temperature profiling at cold spot | HTST processing validation [3] |
| Antibody-based Assays | Specific detection and quantification of proteins | Lactoferrin and IgG quantification in whey proteins [8] |
| Fluorescence Spectroscopy | Assessment of protein structural changes | Whey protein conformation analysis [8] |
| GC-MS Systems | Analysis of volatile compounds and fatty acids | PAH detection and fatty acid profiling [3] [4] |
| Oxygen Radical Absorbance Capacity Assay | Quantification of antioxidant capacity | Evaluation of processed orange juice [7] |
| In vitro Digestion Models | Assessment of bioactive compound bioaccessibility | Carotenoid and phenolic bioaccessibility studies [7] |
The thermal processing paradox presents a complex optimization challenge between ensuring microbial safety and preserving nutritional quality. Thermal technologies, particularly HTST approaches, minimize this paradox through reduced processing times but cannot eliminate nutrient degradation and contaminant formation entirely [3] [4]. Non-thermal technologies demonstrate significant advantages in preserving heat-sensitive bioactive compounds, with documented increases in polyphenol and carotenoid retention, and superior preservation of protein functionality [7] [8]. However, their efficacy varies across food matrices, and optimal application requires matrix-specific parameter optimization. Future research directions should focus on hybrid approaches that combine the safety assurance of thermal processing with the nutrient preservation benefits of non-thermal technologies, while also addressing scale-up challenges and conducting comprehensive life-cycle assessments of these emerging technologies.
In the realm of food science and pharmaceutical development, the processing of bioactive compounds presents a critical paradox: thermal treatments are often essential for ensuring safety and palatability yet can simultaneously degrade nutritional quality and generate harmful compounds. This guide objectively compares the effects of thermal and non-thermal processing on bioactivity, with a specific focus on three fundamental mechanisms of thermal damage: Maillard reactions, vitamin loss, and oxidative changes. The Maillard reaction, a non-enzymatic interaction between reducing sugars and amino groups, is a primary contributor to quality alterations in heat-processed foods, producing both desirable flavors and potentially harmful substances like acrylamide and advanced glycation end-products (AGEs) [9]. Concurrently, heat exposure can degrade heat-sensitive vitamins and accelerate the oxidation of lipids, leading to diminished nutritional value and the formation of off-flavors and toxic compounds [10] [11]. A growing body of research explores non-thermal technologies as alternatives to mitigate these detrimental effects while maintaining microbial safety. This article provides a systematic comparison, supported by experimental data and protocols, to inform researchers and scientists in their selection of processing methods for optimizing bioactivity in functional foods and nutraceuticals.
Comparative Analysis of Thermal Damage Mechanisms
Maillard Reactions: From Flavor Development to Toxic Compounds
The Maillard reaction is a complex network of chemical reactions initiated by the condensation of a carbonyl group (typically from a reducing sugar) and a free amino group (from an amino acid or protein). The reaction proceeds through initial, intermediate, and advanced stages, ultimately yielding a diverse array of molecules collectively known as Maillard reaction products (MRPs) [9]. These products significantly influence the sensory properties, nutritional quality, and safety of processed foods.
Impact on Food Quality and Safety: In the initial stages, MRPs contribute to the development of appealing aromas and flavors, which are desirable in products like bread, roasted coffee, and cooked meats [9]. However, as the reaction progresses into advanced stages, it leads to the formation of brown pigments (melanoidins) and several compounds with documented negative health effects. These include 5-hydroxymethylfurfural (5-HMF), furosine, acrylamide, and heterocyclic amines [12] [9]. Acrylamide, formed from asparagine and reducing sugars, is a neurotoxin and suspected carcinogen [9]. Furthermore, dietary MRPs, particularly AGEs, are associated with an increased risk of chronic diseases such as diabetes, Alzheimer's, and chronic heart failure due to their role in inducing carbonyl stress within the body [9].
Experimental Evidence in Food Systems: The effect of thermal processing on Maillard reaction intensity has been demonstrated across various food matrices. A 2023 study on camel milk showed that the severity of the Maillard reaction escalates with increased heat treatment. When camel milk was heated to temperatures exceeding 120°C, the concentrations of furosine (an indicator of lysine damage) and 5-HMF (an intermediate MRP) increased significantly [12]. Analysis via headspace-gas chromatography-ion mobility spectrometry (HS-GC-IMS) further revealed that the content of aldehydes and ketones—volatile compounds generated during the intermediate stages of the Maillard reaction—also rose noticeably with the intensity of heat treatment [12]. This underscores how thermal processing directly modulates the extent of the Maillard reaction and the profile of its products.
Table 1: Key Maillard Reaction Products and Their Implications
| MRP | Stage of Formation | Effect on Food | Health Implications |
|---|---|---|---|
| Furosine | Initial | Indicator of protein damage & lysine blockage; reduces nutritional value [12] | Not directly toxic, but signifies nutrient loss [12] |
| 5-Hydroxymethylfurfural (5-HMF) | Intermediate | Forms in heated, acidic foods; contributes to color/flavor [12] | Potential carcinogen at high doses [9] |
| Acrylamide | Intermediate (via Strecker deg.) | Forms in starchy foods fried/baked at high temps (>120°C) [9] | Neurotoxin; suspected human carcinogen [9] |
| Melanoidins | Advanced | Brown pigments; contribute to antioxidant activity [9] | May have antioxidant or pro-oxidant effects [9] |
| Advanced Glycation End-products (AGEs) | Advanced | Form in both food and the body during glycation [9] | Linked to diabetes, inflammation, Alzheimer's [9] |
Vitamin Degradation: The Thermolabile Nature of Micronutrients
Vitamins are essential organic micronutrients that are particularly susceptible to degradation during thermal processing. The extent of loss is influenced by the specific vitamin's structure, the processing method, temperature, duration, and the presence of oxygen or light.
Mechanisms of Loss: The primary mechanism for vitamin loss during thermal processing is heat-induced decomposition. Water-soluble vitamins, such as vitamin C and many B vitamins, are especially vulnerable as they can also leach into cooking water [10]. Fat-soluble vitamins (A, D, E, K) are more stable but can degrade through oxidation, particularly when exposed to high temperatures for prolonged periods [10].
Comparative Data on Cooking Methods: A 2017 study investigating the effects of different cooking methods on ten vegetables provides clear quantitative data on vitamin C retention. The study found that microwaving generally led to the highest retention of vitamin C, while boiling caused the most significant losses, with retention rates ranging from 0.0% to 91.1% across the different vegetables [10]. For instance, in broccoli, microwaving preserved a substantially higher percentage of vitamin C compared to boiling. Interestingly, the study also noted that cooking could occasionally increase the content of fat-soluble vitamins like α-tocopherol (vitamin E) and β-carotene (provitamin A), possibly by liberating them from the food matrix, though this effect is highly dependent on the vegetable type [10].
Table 2: Impact of Cooking Methods on Vitamin C Retention in Vegetables [10]
| Vegetable | Boiling | Blanching | Steaming | Microwaving |
|---|---|---|---|---|
| Broccoli | Low | Moderate | Moderate | High |
| Spinach | Low | Moderate | Moderate | High |
| Carrot | Low | Moderate | Moderate | High |
| Potato | Low | Low | Moderate | High |
Oxidative Changes: Compromising Lipid Stability and Bioactivity
Oxidation is a major cause of quality deterioration in foods, particularly in lipids. Thermal processing accelerates oxidative reactions, leading to rancidity, loss of bioactive compounds, and the formation of potentially harmful oxidation products.
Impact on Oils and Bioactive Compounds: Polyunsaturated fatty acids (PUFAs) are highly prone to oxidation, which degrades their nutritional value and generates off-flavors. Thermal treatments, such as the frying of oils at typical temperatures of 170–200°C, induce a series of reactions including hydrolysis, oxidation, and polymerization [11]. These reactions lead to an increase in total polar compounds and triacylglycerol polymers, which are key markers of oil degradation [11]. A 2022 study on oil blends with a balanced ω6/ω3 ratio found that heating at 200°C increased the polar fraction content by an average of 3.1 times compared to unheated oils, indicating significant oxidative damage [11].
Protective Role of Antioxidants and MRPs: The same study highlighted the protective role of natural antioxidants. Tocochromanols (tocopherols and tocotrienols), which constitute vitamin E, act as potent antioxidants by scavenging lipid peroxide radicals. However, they are also degraded during heating. The study reported a dramatic reduction in total tocopherols to just 1–6% of the original content after heating, except in blends containing wheat germ oil, which maintained a high level due to its initially high concentration [11]. Conversely, some MRPs, particularly melanoidins, possess antioxidant properties [9]. Research on thermally treated oilseeds suggests that MRPs extracted along with the oil can contribute to its improved oxidative stability, creating a complex interplay between detrimental and beneficial thermal reaction products [13].
Thermal vs. Non-Thermal Processing: A Data-Driven Comparison
Non-thermal technologies have emerged as promising alternatives to conventional heat treatments, aiming to achieve microbial safety and shelf-life extension with minimal impact on the food's nutritional and sensory qualities.
Direct Impact on Key Damage Mechanisms
Maillard Reaction Control: Non-thermal processes generally result in significantly lower formation of MRPs. For example, freeze-drying of Ginkgo biloba seeds led to substantially lower Maillard reaction product formation (measured as A294 and A420) compared to thermal drying methods like infrared and hot-air drying [14]. Pulsed Electric Fields (PEF) have also been shown to effectively avoid non-enzymatic browning in fruit juices such as strawberry and tomato [9].
Vitamin and Phytochemical Retention: Non-thermal methods excel at preserving heat-labile compounds. High-Pressure Processing (HPP) at 400-600 MPa effectively inactivates enzymes like polyphenol oxidase in potatoes without significantly altering the total phenolic content or antioxidant activity [7]. Similarly, thermo-sonication (a combination of mild heat and ultrasound) in orange juice not only ensured microbial safety but also increased the total polyphenol content by 10.03% and total carotenoids by 20.10%, while largely retaining ascorbic acid and flavonoids [7].
Oxidative Stability: The effect of non-thermal technologies on oxidation is nuanced. While they avoid the pro-oxidative effect of high heat, some technologies can potentially induce oxidative stress. However, many studies report positive outcomes. For instance, the application of PEF to carrots before juicing increased the phenolic bio-accessibility to 100% in purees [7].
Table 3: Comparative Effect of Thermal and Non-Thermal Treatments on Bioactive Compounds
| Processing Technology | Effect on Maillard Reaction | Effect on Vitamins/Phytochemicals | Key Research Findings |
|---|---|---|---|
| Thermal (Pasteurization, UHT) | Significant increase in furosine, HMF, and flavor volatiles [12] | Significant loss of vitamin C; variable effect on fat-soluble vitamins [10] | Camel milk heated at >120°C showed severe Maillard reaction [12] |
| Freeze-Drying (FD) | Lowest MRP formation among tested methods [14] | High retention of volatile compounds and phytochemicals [14] | Ginkgo seeds had lowest A294 (0.582) and least flavor intensity [14] |
| High-Pressure Processing (HPP) | Effectively avoids non-enzymatic browning [9] | No significant change in total phenolics or antioxidant activity in potatoes [7] | In carrot purees, increased phenolic bio-accessibility to 100% [7] |
| Pulsed Electric Fields (PEF) | Avoids non-enzymatic browning in juices [9] | Retains ascorbic acid; can increase bio-accessibility of phenolics [7] | Applied to carrots before juicing, improving compound availability [7] |
| Ultrasound (US) | Can be used to accelerate Maillard conjugation under controlled conditions [15] | In orange juice, increased polyphenols (10%) and carotenoids (20%) [7] | Can be combined with nisin to enhance quality and safety of juice [7] |
Detailed Experimental Protocols
To ensure reproducibility and provide a clear basis for comparison, detailed methodologies from key cited studies are outlined below.
Protocol 1: Analysis of Maillard Reaction Products in Camel Milk [12]
- Objective: To quantify the formation of furosine and 5-HMF in camel milk under different heat treatments (75-135°C for 5-30s).
- Methodology:
- Furosine Analysis: 2 mL of milk was hydrolyzed with 6 mL of 10.6 mol/L HCl at 110°C for 20 hours. The hydrolysate was filtered, mixed with ammonium acetate, and filtered again. Analysis was performed using UPLC with a HSS T3 column and a mobile phase of deionized water with 0.1% trifluoroacetic acid (solvent A) and methanol (solvent B).
- 5-HMF Analysis: 10 mL of milk was mixed with oxalic acid and heated in a boiling water bath for 25 min. Proteins were precipitated using methanol, potassium ferrocyanide, and zinc acetate. The supernatant was filtered and analyzed by UPLC with a C18 column, using methanol and deionized water as the mobile phase.
- Volatile Compounds: Analyzed using Headspace-Gas Chromatography-Ion Mobility Spectrometry (HS-GC-IMS). 2 mL of sample was incubated at 80°C for 20 min, and 500 µL of headspace was injected into the GC-IMS.
Protocol 2: Evaluation of Vitamin Retention in Vegetables [10]
- Objective: To determine the true retention of vitamins (C, E, K, β-carotene) in ten vegetables after blanching, boiling, microwaving, and steaming.
- Methodology:
- Cooking: Vegetables were cooked according to standardized times for each method (e.g., boiling for 5-20 min, microwaving at 700W for 2-5 min). After cooking, samples were drained (if applicable), frozen at -80°C, and lyophilized.
- Vitamin C Analysis: Lyophilized samples were homogenized in a 3% metaphosphoric acid solution. The extract was centrifuged, filtered, and analyzed by HPLC with a CrestPak C18S column and a mobile phase of 0.1% trifluoroacetic acid, with detection at 254 nm.
- Vitamin E Analysis: Samples underwent saponification with ethanolic pyrogallol and potassium hydroxide at 70°C for 50 min. Vitamin E was extracted with n-hexane:ethyl acetate, concentrated under nitrogen, redissolved, and analyzed by normal-phase HPLC with fluorescence detection (ex: 290 nm, em: 330 nm).
Protocol 3: Assessing Oxidative Stability in Oil Blends [11]
- Objective: To analyze the nutritional quality and oxidative stability of oil blends (with a 5:1 ω6/ω3 ratio) during thermal treatment at 170°C and 200°C.
- Methodology:
- Heating Procedure: 50 mL of each oil blend was heated in a thin layer using a steel pan at the target temperatures.
- Tocochromanol Analysis: Content was analyzed before and after heating, likely via HPLC (specific method detailed in referenced patent applications).
- Polar Compound Content: This key marker of oil degradation was measured post-heating. The content increased on average 1.9 and 3.1 times at 170°C and 200°C, respectively.
- Triacylglycerol Polymerization: The formation of dimers and trimers was assessed, with trimers observed particularly at 200°C.
Pathways, Workflows, and Research Tools
Visualization of Key Processes
The following diagrams, generated using Graphviz DOT language, illustrate the core mechanisms and experimental workflows discussed in this guide.
Diagram Title: Maillard Reaction Pathway and Consequences
Diagram Title: Comparative Processing Experimental Workflow
The Scientist's Toolkit: Essential Research Reagents and Materials
This section details key reagents, materials, and instruments essential for conducting research on thermal damage mechanisms, as derived from the experimental protocols cited.
Table 4: Key Research Reagents and Solutions for Thermal Damage Studies
| Item/Reagent | Function in Research | Exemplary Use Case |
|---|---|---|
| Furosine Standard | Quantitative calibration for UPLC/HPLC analysis of early-stage Maillard reaction and lysine blockage [12]. | Determination of heat damage in milk proteins [12]. |
| 5-HMF Standard | Quantitative calibration for UPLC/HPLC analysis of intermediate-stage Maillard reaction [12]. | Monitoring HMF formation in heat-treated camel milk or processed fruits [12]. |
| Trifluoroacetic Acid (TFA) | Ion-pairing reagent in mobile phase for HPLC analysis of polar compounds like furosine [12]. | Used in UPLC mobile phase (0.1% in water) for furosine separation [12]. |
| Metaphosphoric Acid | Protein precipitant and stabilizer for ascorbic acid during extraction, preventing its oxidation [10]. | Extraction of vitamin C from vegetables prior to HPLC analysis [10]. |
| Tocopherol Standards (α, γ, etc.) | Quantitative calibration for HPLC analysis of vitamin E isoforms [10]. | Measuring tocopherol degradation in heated oil blends [11]. |
| Polyphenol Standards (e.g., Gallic Acid, Catechin) | Calibration for spectrophotometric or HPLC determination of total phenolic content and specific polyphenols [16]. | Assessing the impact of thermal vs. non-thermal processing on antioxidant compounds [7]. |
| Headspace Vials / GC-IMS | For capturing and analyzing volatile organic compounds to profile flavor changes and Maillard volatiles [12]. | Detecting increases in aldehydes and ketones in heat-treated camel milk [12]. |
| Electronic Nose (E-nose) | Rapid, non-destructive fingerprinting of overall aroma profile and flavor intensity [14]. | Differentiating flavor profiles of Ginkgo biloba seeds dried by different methods [14]. |
The choice between thermal and non-thermal processing methods presents a significant trade-off for researchers and industry professionals. Thermal processing is a robust and well-established technology for ensuring microbial safety and developing desired sensory attributes. However, this review has quantitatively demonstrated its drawbacks: the inevitable induction of Maillard reactions (with associated nutrient loss and toxicant formation), significant degradation of heat-labile vitamins, and the acceleration of oxidative processes in lipids. In contrast, non-thermal technologies such as HPP, PEF, and ultrasound offer a compelling alternative, demonstrating superior retention of vitamins and phytochemicals and a marked reduction in the formation of harmful MRPs. The experimental data confirms that these technologies can effectively preserve, and in some cases even enhance, the bioactivity and nutritional quality of food matrices.
The decision-making process must be application-specific. For products where a cooked flavor and color are desirable, mild and controlled thermal processing may be appropriate. However, for the preservation of maximum nutritional value and the minimization of processing contaminants, non-thermal technologies are demonstrably more effective. Future research should focus on optimizing these non-thermal technologies for industrial scale-up, conducting more long-term stability studies on processed products, and deepening the understanding of synergistic effects when these technologies are combined with mild heat or other hurdles. This evidence-based comparison provides a foundation for making informed decisions that align with the goals of developing safer, healthier, and more bioactive food and nutraceutical products.
Non-thermal preservation technologies represent a transformative approach to microbial inactivation in food and biological materials. Unlike conventional thermal processing, which relies on heat to destroy microorganisms but often degrades heat-sensitive nutrients and bioactive compounds, non-thermal methods operate at or near ambient temperatures [17] [5]. This fundamental difference allows for the selective targeting of microbial structures while sparing valuable molecular components, making these technologies particularly valuable for preserving bioactivity in functional foods, pharmaceuticals, and nutraceuticals [2]. The growing consumer demand for high-quality, minimally processed foods with fresh-like characteristics has accelerated the development and implementation of these technologies across various sectors of the food industry [18]. This review comprehensively compares the mechanisms, efficacy, and applications of major non-thermal technologies, with particular emphasis on their capacity to maintain bioactive compound integrity while ensuring microbial safety.
Comparative Mechanisms of Microbial Inactivation
Fundamental Principles
Non-thermal technologies inactivate microorganisms through physical or chemical mechanisms that selectively target cellular structures without significantly elevating temperature. The primary cellular targets include membranes, enzymes, and genetic material, with specific mechanisms varying by technology [19] [20]. Table 1 summarizes the primary inactivation mechanisms and cellular targets for each major non-thermal technology.
Table 1: Microbial Inactivation Mechanisms of Non-Thermal Technologies
| Technology | Primary Mechanism | Cellular Targets | Critical Process Parameters |
|---|---|---|---|
| High Pressure Processing (HPP) | Isostatic pressure application; follows Le Chatelier's principle [21] [17] | Cell membrane integrity, key enzymes, protein conformation [20] | Pressure (100-800 MPa), holding time, temperature [21] |
| Pulsed Electric Field (PEF) | Electroporation and electrical breakdown of cell membranes [17] | Cytoplasmic membrane, intracellular components [17] | Field strength (10-80 kV/cm), pulse width, specific energy [17] |
| Cold Plasma (CP) | Reactive oxygen and nitrogen species (RONS) action [2] | Cell membrane, enzymes, genetic material [20] | Gas composition, power input, exposure time [2] |
| Ultrasound (US) | Cavitation-induced shear forces and localized heating [5] | Cell walls, membranes [5] | Frequency (20-100 kHz), amplitude, treatment time [5] |
| Pulsed Light (PL) | Photochemical and photothermal damage [22] | DNA, cellular components [22] | Wavelength, fluency, pulse duration [22] |
| Ultraviolet (UV) | DNA damage via thymine dimer formation [2] | Genetic material [2] | Intensity, exposure time, product transparency [2] |
| Ozonation | Strong oxidative action on cellular components [2] | Cell membranes, enzymes [2] | Concentration, contact time, humidity [2] |
Microbial Inactivation Pathways
The following diagram illustrates the general microbial inactivation pathways common to multiple non-thermal technologies, highlighting how these processes target microorganisms while minimizing damage to food components.
Quantitative Comparison of Processing Technologies
Microbial Efficacy Across Technologies
Different non-thermal technologies exhibit varying efficacy against microorganisms, influenced by processing parameters, food matrix, and microbial characteristics. Table 2 provides a comparative overview of inactivation capabilities against common microorganisms.
Table 2: Microbial Inactivation Efficacy of Non-Thermal Technologies
| Technology | Target Microorganisms | Reduction Range (log CFU) | Key Influencing Factors |
|---|---|---|---|
| HPP | Vegetative bacteria, molds, yeasts, viruses [21] | 3-6 log for most vegetative cells [21] | Pressure level, holding time, temperature, food composition [21] |
| PEF | Vegetative bacteria, yeasts [17] | 3-5 log in liquid foods [17] | Field strength, pulse number, conductivity, temperature [17] |
| Cold Plasma | Broad spectrum: bacteria, molds, yeasts, spores [2] | 1-5 log depending on exposure [2] | Gas composition, power, treatment time, sample distance [2] |
| Ultrasound | Bacteria, yeasts (often with hurdles) [5] | 1-3 log (higher with combination) [5] | Frequency, amplitude, time, temperature, food matrix [5] |
| Pulsed Light | Surface microorganisms, viruses [22] | 2-5 log on surfaces [22] | Fluence, pulse duration, transparency, surface geometry [22] |
| UV | Surface bacteria, molds, viruses [2] | 1-4 log depending on exposure [2] | Intensity, exposure time, penetration depth [2] |
| Ozonation | Broad spectrum including spores [2] | 1-5 log depending on application [2] | Concentration, contact time, humidity, organic matter [2] |
Impact on Bioactive Compounds
The preservation of bioactive compounds represents a significant advantage of non-thermal technologies over thermal processing. Table 3 compares the effects of different technologies on heat-sensitive nutrients and bioactive compounds.
Table 3: Impact on Bioactive Compounds Compared to Thermal Processing
| Technology | Vitamin Retention | Phytochemical Preservation | Overall Bioactivity |
|---|---|---|---|
| Thermal Processing | Significant loss of heat-sensitive vitamins (e.g., Vitamin C) [5] | Degradation of thermolabile compounds; Maillard reactions [18] | Substantial reduction in antioxidant capacity [23] |
| HPP | Excellent retention (>90%) of vitamins [2] | Enhanced extraction and stability of polyphenols [2] | Well-preserved or enhanced bioactivity [2] |
| PEF | High retention of heat-sensitive vitamins [2] | Improved release and stability of bioactive compounds [17] | Maintained or slightly improved antioxidant activity [17] |
| Cold Plasma | Variable effects depending on exposure [2] | May degrade some surface compounds due to oxidation [18] | Context-dependent; can preserve or moderately reduce [2] |
| Ultrasound | Good retention of vitamins [5] | Enhanced extraction of intracellular compounds [5] | Often improved bioavailability and activity [5] |
| UV | Potential degradation of light-sensitive vitamins [2] | Generally good preservation of most compounds [2] | Well-maintained with proper dosing [2] |
| Ozonation | Good retention of most vitamins [2] | May oxidize some sensitive phytochemicals [2] | Generally maintained with minimal processing [2] |
Experimental Protocols for Key Technologies
High-Pressure Processing Experimental Protocol
Principle: HPP follows Le Chatelier's principle and the isostatic rule, where pressure is uniformly distributed throughout the product regardless of its geometry [21] [17]. Pressure affects non-covalent bonds (hydrogen, ionic, and hydrophobic), leading to protein denaturation, enzyme inactivation, and membrane disruption in microorganisms [20].
Detailed Methodology:
- Sample Preparation: Package food samples in flexible, water-resistant packaging (pouches or plastic bottles) to allow pressure transmission [21].
- Loading: Place packaged samples in the pressure vessel filled with pressure-transmitting fluid (typically water) [21].
- Pressure Application: Close vessel and pump additional fluid to achieve target pressure (typically 300-600 MPa for microbial inactivation) [21].
- Holding Time: Maintain pressure for specified duration (1-10 minutes depending on application) [21].
- Depressurization: Rapidly release pressure and remove samples [21].
- Analysis: Assess microbial counts, nutrient retention, and quality parameters immediately after treatment and throughout storage [21].
Critical Parameters:
- Pressure level: 100-800 MPa (typically 400-600 MPa for pasteurization) [21]
- Holding time: Seconds to 20 minutes [21]
- Temperature: Ambient or moderate (<45°C) [21]
- Come-up time: Varies by equipment [21]
- Product characteristics: pH, water activity, composition [21]
Pulsed Electric Field Experimental Protocol
Principle: PEF technology applies short, high-voltage pulses (typically 10-80 kV/cm for microseconds) to liquid foods placed between two electrodes [17]. The electric field causes electroporation, forming pores in microbial membranes, which leads to irreversible cell damage and death when severe [17].
Detailed Methodology:
- Sample Preparation: Ensure homogeneous liquid food with controlled electrical conductivity [17].
- System Setup: Configure PEF system with treatment chamber, high-voltage pulse generator, fluid handling system, and control units [17].
- Processing: Pump food through treatment chamber while applying high-voltage pulses at specific field strength, pulse width, and frequency [17].
- Temperature Control: Use cooling system to maintain temperature below thermal damage threshold (typically <40°C) [17].
- Collection: Collect treated sample aseptically for analysis [17].
Critical Parameters:
- Electric field strength: 10-80 kV/cm [17]
- Pulse width: 1-10 microseconds [17]
- Total treatment time: Microseconds to milliseconds [17]
- Pulse waveform: Exponential decay or square wave [17]
- Food properties: Conductivity, pH, composition [17]
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Reagent Solutions for Non-Thermal Processing Studies
| Reagent/Material | Function in Research | Application Examples |
|---|---|---|
| Selective Culture Media | Differentiation between injured and viable cells [19] | Detection of sublethally injured microorganisms after HPP, PEF [19] |
| Viability Stains (FDA, PI) | Cell viability assessment via membrane integrity [19] | Distinguishing live/dead cells after non-thermal treatments [19] |
| ATP Measurement Kits | Metabolic activity quantification [19] | Detection of VBNC cells that retain metabolic activity [19] |
| PCR/DNA Extraction Kits | Genetic damage assessment and pathogen detection [19] | Confirming genetic material damage after PL, UV treatments [19] |
| Antioxidant Assay Kits | Quantification of oxidative stress in microbes and foods [18] | Evaluating efficacy of oxidative technologies (CP, ozone) [2] |
| Protein Analysis Reagents | Protein denaturation and structural change assessment [18] | Studying protein modifications after HPP, PEF treatments [18] |
| HPLC Standards | Bioactive compound quantification [23] | Vitamin, polyphenol retention after non-thermal processing [23] |
Hidden Risks: Sublethal Injury and VBNC State
A significant research consideration in non-thermal processing is the potential induction of sublethal injury and the viable but non-culturable (VBNC) state in microorganisms [19] [20]. In the VBNC state, bacteria maintain metabolic activity and potential pathogenicity but cannot form colonies on standard culture media, leading to underestimation of survival risks [19]. Multiple non-thermal technologies have demonstrated this capacity:
- HPP may cause reversible damage to cell structures rather than complete inactivation [19]
- PEF can induce sublethal membrane damage that may be repaired under favorable conditions [19]
- Cold plasma and ozonation may trigger oxidative stress responses that promote VBNC state [20]
The following diagram illustrates the transition pathways to the VBNC state and potential resuscitation mechanisms following non-thermal treatments.
Detection of VBNC cells requires specialized methods beyond conventional plating, including:
- Direct viable count with viability stains [19]
- Molecular methods (qPCR, RNA-based detection) [19]
- Metabolic activity assays (ATP measurement, respiration indicators) [19]
Non-thermal preservation technologies offer compelling alternatives to thermal processing by effectively inactivating microorganisms while preserving valuable bioactive compounds. Each technology presents unique mechanisms and applications, with HPP and PEF being particularly effective for liquid and semi-solid foods while maintaining nutrient integrity [2]. However, researchers must consider potential challenges including sublethal injury, VBNC state induction, and variable effects on different food matrices [19] [20]. The selection of appropriate non-thermal technology depends on the specific food product, target microorganisms, desired shelf life, and critical quality attributes. Future research should focus on optimizing processing parameters, understanding recovery mechanisms of injured cells, and developing detection methods for comprehensive microbial risk assessment. When properly applied, non-thermal technologies represent powerful tools for developing safer, more nutritious products with enhanced bioactivity.
In the pursuit of developing functional foods and nutraceuticals, researchers and drug development professionals must navigate the critical impact of food processing on bioactive compounds. Bioactive compounds in fruits and vegetables, including polyphenols, flavonoids, anthocyanins, and vitamins, exert positive effects on human health by reducing oxidative stress, inflammation, and the risk of chronic diseases such as cancer, cardiovascular diseases, and metabolic disorders [24]. The efficacy of these bioactives depends not only on their initial content but also on their stability during processing and their ultimate bioaccessibility in the human body [25]. This guide provides a comprehensive comparison of how thermal and non-thermal processing technologies affect the retention and bioactivity of these crucial compounds, supported by experimental data and mechanistic insights.
Comparative Impact of Processing Technologies on Key Bioactives
The selection of processing method significantly influences the stability, retention, and bioavailability of bioactive compounds. The following sections and comparative tables synthesize findings from recent studies on thermal and non-thermal processing effects.
Polyphenols and Flavonoids
Polyphenols, which include flavonoids, phenolic acids, lignans, and stilbenes, are widely distributed in the plant kingdom and recognized for their antioxidant and anti-inflammatory properties [24]. Processing can significantly alter their content and bioaccessibility.
Table 1: Effects of Processing on Total Phenolic Content (TPC) and Total Flavonoid Content (TFC)
| Processing Technology | Specific Conditions | Matrix | Effect on TPC/TFC | Key Findings |
|---|---|---|---|---|
| Pulsed Electric Field (PEF) | 120 kJ/L, 24 kV/cm | Fruit juice blend (kiwi, mango, orange, blueberry) | ↑↑ Highest bioactive content post-processing [26] | Highest phenolic bioaccessibility after in vitro digestion [26] |
| High-Pressure Processing (HPP) | 600 MPa, 3 min | Fruit juice blend (kiwi, mango, orange, blueberry) | ↑↑ Highest bioactive content post-processing [26] | Bioactive content protected during storage, similar to thermal treatment [26] |
| Cold Plasma (CP) | 50-60 kV, 5-10 min | Buckwheat grain and flour | ↑ Significant increase | TPC increased to 83.99 mg GAE/g DW; TFC to 96.60 mg RE/g DW [27] |
| Thermal Treatment (TT) | 80°C, 30 min | Fruit juice blend | ↓ Variable | Bioactive content protected during storage, but lower bioaccessibility vs. PEF [26] |
| Boiling | 100°C, variable times | Various vegetables (e.g., tatsoi, artichoke) | ↓↓ Significant decrease (e.g., -27.1% TPC in tatsoi) | Loss due to leaching into water and thermal degradation [24] |
Anthocyanins
Anthocyanins, responsible for red, blue, and purple pigmentation in plants, are particularly sensitive to environmental conditions. Their stability is highly dependent on processing parameters and the food matrix [28].
Table 2: Effects of Processing on Anthocyanins and Antioxidant Activity
| Processing Technology | Specific Conditions | Matrix | Effect on Anthocyanins & Antioxidant Activity | Key Findings |
|---|---|---|---|---|
| Pulsed Electric Field (PEF) | 120 kJ/L, 24 kV/cm | Fruit juice blend | ↑ Highest total anthocyanin content (TAC) post-digestion [26] | Enhanced bioaccessibility and retention of antioxidant capacity [26] |
| High-Pressure Processing (HPP) | 600 MPa, 3 min | Fruit juice blend | ↑ High initial content | Individual anthocyanin content decreased during storage [26] |
| Cold Plasma (CP) | 50-60 kV, 5-10 min | Buckwheat | ↑↑ Significant increase in antioxidant activity | DPPH radical scavenging activity up to 92.25%; FRAP value 48.09 mmol Fe²⁺/mg DW [27] |
| Ultra High Temperature (UHT) | Not specified | Açai juice | ↑ Anthocyanin bioaccessibility | 2-fold higher anthocyanin bioaccessibility post-processing [29] |
| Thermal Treatment | General heating | Anthocyanin extracts & foods | ↓↓ Degradation dependent on time/temperature | Degradation follows Arrhenius kinetics; complex interaction with food matrix can offer protection [28] |
Vitamins and Heat-Sensitive Compounds
Vitamins, particularly vitamin C (ascorbic acid), are highly susceptible to degradation under heat and other environmental stressors.
Table 3: Effects of Processing on Vitamins and Heat-Sensitive Proteins
| Processing Technology | Specific Conditions | Matrix | Effect on Vitamins & Sensitive Compounds | Key Findings |
|---|---|---|---|---|
| Ultrasound + LPP | Combined treatment | Açai juice | ↑ Vitamin C bioaccessibility | 8% improvement in vitamin C bioaccessibility [29] |
| Thermo-sonication + Nisin | Combined treatment | Orange juice | = Ascorbic acid retained | Flavonoids and ascorbic acid largely retained [7] |
| High-Temperature Long-Time (HTLT) | Excessive heat | Donkey milk whey | ↓↓↓ Severe loss of active proteins | Lysozyme activity preserved only 23%; Lactoferrin and IgG losses of 70% and 77% [8] |
| Ultrasonication / UV | Non-thermal | Donkey milk whey | ↑ Superior preservation of proteins | Better preservation of lysozyme, lactoferrin, and IgG compared to heat [8] |
| Ultra High Temperature (UHT) | Not specified | Açai juice | ↓↓↓ Sharp decrease | Vitamin C bioaccessibility sharply decreased [29] |
Experimental Protocols for Assessing Bioactive Stability
To generate comparable data on bioactive stability, standardized experimental protocols are essential. Below are methodologies commonly used in the cited studies.
In Vitro Digestion Model
This protocol simulates the human gastrointestinal tract to evaluate the bioaccessibility of bioactive compounds after processing [26] [29].
- Oral Phase: The processed sample is mixed with simulated salivary fluid and incubated for a few minutes.
- Gastric Phase: The oral bolus is mixed with simulated gastric fluid containing pepsin, and the pH is adjusted. The mixture is incubated at 37°C for a set period (e.g., 1-2 hours) with constant agitation.
- Intestinal Phase: The gastric chyme is mixed with simulated intestinal fluid containing pancreatin and bile salts. The pH is adjusted to neutral, and the mixture is incubated further at 37°C.
- Analysis: The digestate is centrifuged. The supernatant (bioaccessible fraction) is collected and analyzed for target bioactives (e.g., TPC, TFC, TAC, specific vitamins) using spectrophotometric, HPLC, or other techniques.
Analysis of Total Phenolic and Flavonoid Content
These are standard colorimetric assays for quantifying two major classes of polyphenols [27].
- Total Phenolic Content (TPC) by Folin-Ciocalteu Assay: The sample extract is mixed with Folin-Ciocalteu reagent and sodium carbonate. After incubation, the absorbance of the resulting blue complex is measured at 765 nm. Results are expressed as gallic acid equivalents (mg GAE/g) [27].
- Total Flavonoid Content (TFC) by Aluminum Chloride Method: The sample extract is mixed with sodium nitrite and aluminum chloride. After the addition of sodium hydroxide, the absorbance of the pink complex is measured at 510 nm. Results are expressed as rutin equivalents (mg RE/g) [27].
Antioxidant Activity Assays
Multiple assays are used to probe different antioxidant mechanisms.
- DPPH Radical Scavenging Activity: The sample is mixed with a methanolic solution of the stable DPPH radical. The decrease in absorbance at 517 nm is measured after incubation in the dark. The percentage of radical scavenging activity is calculated [27].
- FRAP (Ferric Reducing Antioxidant Power): The sample is mixed with the FRAP reagent (TPTZ solution, FeCl₃ solution, and acetate buffer). The increase in absorbance at 593 nm, due to the formation of a blue-colored Fe²⁺-TPTZ complex, is measured. Results are expressed as mmol Fe²⁺ equivalents [27].
- ORAC (Oxygen Radical Absorbance Capacity): The sample is mixed with a fluorescent probe and an oxygen radical generator (AAPH). The fluorescence decay is monitored over time. The area under the curve is compared to a Trolox standard, and results are expressed as μmol TE (Trolox Equivalents) [30].
Mechanisms of Action: How Processing Technologies Affect Bioactives
The following diagrams illustrate the mechanistic pathways through which thermal and non-thermal processing technologies impact the stability, content, and bioavailability of bioactive compounds in food matrices.
Non-Tthermal Processing Mechanism
Thermal Processing Impact Pathway
Anthocyanin Degradation Mechanism
The Scientist's Toolkit: Key Research Reagents and Materials
This table details essential reagents, materials, and equipment used in the experiments cited throughout this guide, providing a reference for researchers seeking to replicate or design similar studies.
Table 4: Essential Research Reagents and Materials for Bioactive Analysis
| Reagent / Material / Equipment | Functional Role in Research | Example Application |
|---|---|---|
| Folin-Ciocalteu Reagent | Colorimetric detection and quantification of total phenolic compounds via redox reaction. | Total Phenolic Content (TPC) assay [27]. |
| DPPH (1,1-diphenyl-2-picrylhydrazyl) | Stable free radical used to evaluate the free radical scavenging capacity of antioxidant compounds. | DPPH radical scavenging assay [27]. |
| TPTZ (2,4,6-Tripyridyl-s-triazine) | Chromogenic compound that complexes with ferrous ions to measure the reducing power of antioxidants. | FRAP (Ferric Reducing Antioxidant Power) assay [27]. |
| Simulated Gastrointestinal Fluids | Chemically defined solutions mimicking saliva, gastric, and intestinal juices for in vitro digestion studies. | Assessment of bioaccessibility after processing [26]. |
| HPLC-Grade Standards (e.g., Rutin, Quercetin, Gallic Acid) | High-purity reference compounds for calibration curves and identification/quantification of specific bioactives. | Quantification of individual flavonoids and phenolic acids [27]. |
| High-Pressure Processing (HPP) Unit | Industrial-scale equipment applying isostatic pressure (100-600 MPa) for cold pasteurization. | Studying HPP effects on juice bioactives [26] [2]. |
| Dielectric Barrier Discharge (DBD) Plasma Reactor | Device generating cold plasma at atmospheric pressure for surface decontamination and material modification. | Treating buckwheat grain and flour [27]. |
| Pulsed Electric Field (PEF) System | Equipment applying short, high-voltage pulses to permeabilize cell membranes. | PEF treatment of fruit juices [26] [2]. |
Non-Thermal Technologies in Action: Mechanisms and Food System Applications
Consumer demand for fresh-like, nutritious, and minimally processed foods has driven the development of non-thermal preservation technologies as alternatives to conventional heat treatments. High-Pressure Processing (HPP) has emerged as a leading non-thermal technology, capable of ensuring microbial safety while better preserving heat-sensitive nutritional components compared to thermal pasteurization. This guide provides an objective comparison of HPP and thermal processing, focusing on their mechanisms and differential impacts on the bioactive compounds in fruits and vegetables, crucial for researchers and scientists in food science and product development.
Fundamental Mechanisms of HPP
HPP employs hydrostatic pressure, typically in the range of 100 to 600 MPa, transmitted uniformly and instantaneously by a pressure-transmitting medium (usually water) to packaged food products [31] [2]. Its effectiveness stems from foundational physical-chemical principles:
- Isostatic Principle: Pressure is distributed uniformly throughout the product, independent of its geometry, ensuring homogeneous treatment [31] [32].
- Le Chatelier’s Principle: Any phenomenon in equilibrium (e.g., biochemical reaction, molecular configuration) accompanied by a volume decrease is enhanced by pressure application [31] [32].
- Microscopic Ordering Principle: Increased pressure raises the degree of molecular ordering of a substance at constant temperature [32].
These principles govern HPP's effects on microbial and food matrices, while its ability to preserve bioactives arises from its minimal impact on low molecular weight covalent bonds, which form the structure of vitamins, pigments, and antioxidants [31] [33].
Mechanisms of Microbial and Enzyme Inactivation
HPP inactivates microorganisms primarily through pressure-induced damage to cell membranes, protein denaturation, and enzyme inactivation [34]. Gram-negative bacteria are generally more pressure-sensitive than Gram-positive bacteria due to differences in cell wall structure [34]. The efficacy depends on pressure level, holding time, temperature, and food composition [35]. Endogenous enzymes in fruits and vegetables, such as polyphenol oxidase (PPO) and pectin methylesterase (PME), can be baro-resistant, sometimes requiring combined pressure-thermal treatments for full inactivation [36].
Comparative Analysis: HPP vs. Thermal Processing on Bioactives
The following sections and tables synthesize experimental data comparing the effects of HPP and thermal processing on key bioactive compounds.
Impact on Antioxidant Vitamins
Table 1: Effects of HPP and Thermal Processing on Antioxidant Vitamins in Fruit/Vegetable Products
| Product Matrix | Treatment Conditions | Vitamin C (Ascorbic Acid) | Vitamin A / Carotenoids | Key Findings | Citation |
|---|---|---|---|---|---|
| Fruit Juice Blend | HPP: 600 MPa/3 minTT: 80°C/30 min | Higher retention with HPP | Better retention of total carotenoids with HPP | HPP at 600 MPa/3 min showed the highest bioactive content and antioxidant capacity. | [26] |
| Clementine Juice | HPP: 400 MPa/40°C/1 min | N/A | Total carotenoids concentration higher in HPP | HPP treatment resulted in better stability and bio-accessibility of carotenoids after in vitro digestion. | [33] |
| Wheatgrass Juice | HPP: Not SpecifiedTT: Not Specified | N/A | N/A | HPP prevented color loss and increased chlorophyll content versus thermal treatment. | [37] |
| Strawberry Juice | HPP: 600 MPa/3 minTP: 85°C/2 min | Significant loss in both after storage | N/A | Total loss of ascorbic acid occurred in both HPP and TP samples during storage. | [36] |
Impact on Phenolic Compounds and Antioxidant Capacity
Table 2: Effects of HPP and Thermal Processing on Phenolics and Antioxidant Activity
| Product Matrix | Treatment Conditions | Total Phenolics/ Flavonoids | Anthocyanins / Betanin | Antioxidant Capacity | Citation |
|---|---|---|---|---|---|
| Djulis Whole Grain | HPP: 500 MPa/10-30°C/3 minThermal: Cooking (20 min), Pasteurization (65°C/30 min) | Retained >85% of betanin (major pigment) | HPP retained >85% of betanin, while thermal methods caused significant degradation. | Higher antioxidant capacity in HPP samples compared to thermally processed ones. | [38] |
| Strawberry Juice | HPP: 500 MPa/1.5 or 3 minTP: 85°C/2 min | N/A | Anthocyanins decreased significantly during storage in HPP | HPP sample at 600 MPa/3 min had significantly lower antioxidant activity. | [36] |
| Fruit Juice Blend | HPP: 600 MPa/3 minPEF: 120 kJ/L-24 kV/cmTT: 80°C/30 min | Highest content with HPP (600 MPa/3 min) and PEF (120 kJ/L-24 kV/cm) | N/A | Highest antioxidant capacity with HPP (600 MPa/3 min) and PEF (120 kJ/L-24 kV/cm). | [26] |
| Chickpeas | HPP: 600 MPa/5 min | Polyphenol content and antioxidant activities decreased | N/A | Sample at highest pressure/longest duration (600 MPa/5 min) showed the lowest values. | [35] |
Impact on Color and Sensory Properties
Color is a critical quality attribute directly influenced by pigment stability. HPP's minimal impact on covalent bonds helps preserve chlorophylls in green vegetables [33], anthocyanins in berries [33], and betanin in Djulis [38] more effectively than thermal processing. Studies on strawberry products report that HPP better maintains fresh-like sensory attributes, while thermal processing often leads to cooked flavors and color degradation [36] [31].
Detailed Experimental Protocols for Key Studies
- Sample Preparation: Australian kabuli chickpeas were soaked overnight, cooked in boiling water for 30 minutes, vacuum-packaged, and stored at 4°C until HPP.
- HPP Treatment: Cooked chickpeas were processed at 200, 400, and 600 MPa for 1 and 5 minutes at 4°C using commercial HPP equipment (Model: Hyperbaric 300). Pressure come-up times were 60, 130, and 240 seconds for 200, 400, and 600 MPa, respectively, with instant decompression.
- Analysis:
- Texture Profile Analysis (TPA): Using a texture analyzer with 50% compression, a 5 kg load cell, and 40 replicates.
- Starch Digestibility: Classified into rapidly digestible (RDS), slowly digestible (SDS), and resistant (RS) starch using enzymatic methods.
- Total Polyphenol Content & Antioxidant Activities: Polyphenols extracted with 50% acetone, measured with Folin-Ciocalteu reagent. Antioxidant activities determined via DPPH, ABTS, and ORAC assays.
- Sample Preparation: Red Djulis whole grains, hull, and de-hulled seeds were prepared. Samples (20 g) were mixed with water (40 mL) and aseptically packaged.
- Treatment Conditions:
- HPP: 500 MPa at 10°C and 30°C for 3 minutes.
- Thermal Methods: Pasteurization (65°C for 30 minutes) and cooking (boiling for 20 minutes).
- Analysis:
- Microbial Load: Total plate count.
- Betanin Content: Analyzed using HPLC.
- Color: Measured using a colorimeter.
- Antioxidant Capacities: Assessed via DPPH scavenging activity, FRAP, and SOD-like assays.
- Statistical Analysis: Correlation and Principal Component Analysis (PCA) were performed.
Mechanisms and Workflow Visualization
The following diagram illustrates the core mechanisms of HPP and its contrasting effects on food components compared to thermal processing.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for HPP Bioactivity Research
| Item | Function / Application | Exemplary Use Case |
|---|---|---|
| Total Starch Assay Kit (AA/AMG) | Quantification of total starch content and classification into starch digestibility fractions (RDS, SDS, RS). | Used in chickpea study to analyze starch digestibility [35]. |
| Folin-Ciocalteu Reagent | Measurement of total polyphenol content (TPC) in plant extracts via colorimetric assay. | Standard method for TPC determination in chickpea and fruit juice studies [35] [26]. |
| DPPH, ABTS, FRAP Reagents | Standardized reagents for determining antioxidant capacity through various mechanisms (radical scavenging, reduction power). | Used to assess antioxidant activity in chickpea, Djulis, and fruit juice studies [35] [26] [38]. |
| HPLC Standards | High-Performance Liquid Chromatography standards for quantifying specific bioactive compounds (e.g., betanin, anthocyanins, vitamins). | Essential for precise quantification of individual pigments in Djulis and vitamins in fruit juices [38] [32]. |
| In Vitro Digestion Models | Simulated gastrointestinal fluids to study bioaccessibility of bioactive compounds after processing. | Used to evaluate carotenoid bioaccessibility in clementine juice and phenolic bioaccessibility in fruit juice blends [26] [33]. |
The consolidated experimental data demonstrates that HPP is a superior technology for preserving heat-sensitive bioactive compounds, pigments, and antioxidant activities in fruit and vegetable products compared to traditional thermal processing. While HPP effectively ensures microbial safety, its limitations concerning certain enzyme activations and potential bioactive degradation at very high pressures warrant consideration. The choice between HPP and thermal processing ultimately depends on the target product's specific requirements, balancing the desired shelf-life, nutritional quality, sensory attributes, and economic feasibility. For research and development focused on maximizing the health-promoting potential of plant-based foods, HPP presents a compelling non-thermal alternative.
Pulsed Electric Field (PEF) technology represents a significant advancement in non-thermal food processing, offering an effective method for microbial inactivation while preserving the nutritional and sensory qualities of liquid foods. This technology utilizes short bursts of high voltage electricity to disrupt the cell membranes of microorganisms, thereby achieving preservation without the extensive use of heat that characterizes conventional thermal pasteurization. The growing consumer demand for fresh-like, minimally processed foods with high nutritional value has driven considerable interest in PEF technology within the food industry and research community [39] [40]. As a non-thermal preservation method, PEF aligns with modern sustainability goals through its reduced energy consumption and minimal use of chemical additives [2].
The fundamental principle underlying PEF technology is electroporation, where the application of an external electric field induces pore formation in cell membranes. When the electric field strength exceeds a critical threshold, typically between 15-40 kV/cm for microbial cells, these pores become irreversible, leading to cell death [40]. This mechanism allows PEF to effectively inactivate vegetative bacteria, yeasts, and molds in various liquid food matrices, including fruit juices, milk, and liquid eggs [39] [41]. Unlike thermal processing, which acts through protein denaturation, PEF specifically targets cell membrane integrity, thereby minimizing damage to heat-sensitive food components such as vitamins, antioxidants, and flavor compounds [42].
Fundamental Principles of PEF
Mechanism of Action: Electroporation
The core mechanism of PEF technology is electroporation, a physical process that involves the application of external electric fields to biological cells, resulting in the formation of pores in cell membranes. When a microbial cell is exposed to an electric field of sufficient strength, the transmembrane potential increases, creating electrostatic pressure that opposes the membrane's natural bilayer structure. This pressure causes the phospholipid molecules to reorient themselves, forming hydrophilic pores that compromise the membrane's semi-permeability [39] [40]. The diagram below illustrates this electroporation process and its consequences for microbial inactivation.
The effectiveness of electroporation depends on several factors, including electric field strength, pulse characteristics, treatment temperature, and the properties of the target microorganisms. Gram-negative bacteria are generally more sensitive to PEF than Gram-positive bacteria due to differences in their cell wall structure. Bacterial spores, with their protective coats, are highly resistant to PEF and require combination with other preservation methods for effective inactivation [39].
Critical Processing Parameters
The microbial inactivation efficacy of PEF treatment depends on several interconnected parameters that must be carefully controlled:
- Electric Field Strength: Typically ranges from 20-40 kV/cm for microbial inactivation. Higher field strengths generally increase inactivation rates but also elevate energy costs and potential heating effects [42] [40].
- Treatment Time and Pulse Characteristics: Total treatment time is determined by pulse number, duration, and frequency. Pulse widths typically range from 1 to 10 microseconds, with frequencies from 1 to hundreds of Hz [42].
- Pulse Waveforms: Exponential decay and square wave pulses are most common, with square waves generally being more energy-efficient [40].
- Treatment Temperature: Although PEF is considered non-thermal, some temperature increase occurs due to ohmic heating. Moderate pre-heating can synergistically enhance microbial inactivation [40].
- Product Properties: Electrical conductivity, pH, and composition significantly influence PEF effectiveness. Products with higher electrical conductivity require higher energy input to achieve the same field strength [40].
PEF in Context: Comparison with Alternative Technologies
PEF Versus Conventional Thermal Processing
The comparison between PEF and conventional thermal processing reveals significant differences in their approaches to microbial safety and quality preservation. The table below summarizes key comparative aspects based on experimental findings.
Table 1: Comparative Analysis of PEF vs. Conventional Thermal Processing
| Parameter | PEF Processing | Conventional Thermal Processing |
|---|---|---|
| Microbial Inactivation | 5-log reduction achieved at 30 kV/cm with 10 pulses [42] | Effective across most microorganisms but requires high temperatures (70-100°C) [42] |
| Energy Consumption | Up to 50% reduction compared to thermal methods [43] | High energy requirements for heating and cooling [43] |
| Vitamin C Retention | Significantly higher retention in orange juice [42] | Substantial degradation due to heat sensitivity [42] |
| Color Preservation | Minimal impact on pigments; maintains fresh-like appearance [42] [44] | Often causes browning or other color changes [44] |
| Sensory Properties | Well-preserved fresh-like flavor and aroma [39] [41] | Cooked flavors and aroma loss common [39] |
| Processing Time | Rapid, continuous processing (microseconds to milliseconds) [43] | Longer processing including heating, holding, and cooling [43] |
The superior nutrient retention of PEF is particularly evident in studies with vitamin C-rich products. Research on Thai orange juice demonstrated that PEF treatment preserved significantly more vitamin C compared to conventional thermal pasteurization conducted at 68.2°C for 30 minutes [42]. Similarly, PEF-treated juices maintained higher levels of bioactive compounds such as polyphenols and carotenoids, which are often degraded by thermal processing [7].
PEF Versus Other Non-Thermal Technologies
Among non-thermal technologies, High Pressure Processing (HPP) represents the most established alternative to PEF. The table below compares their performance based on published studies.
Table 2: Comparison of PEF and HPP in Juice Processing
| Characteristic | PEF Technology | High Pressure Processing (HPP) |
|---|---|---|
| Microbial Stability | Microbial regrowth after 28 days in strawberry juice [44] | Maintains microbial counts below 2 log CFU/mL for 42+ days [44] |
| Enzyme Inactivation | Effective with outlet temperatures near 73°C; 34% PME inactivation in orange juice [44] | Variable effectiveness; 92% PME inactivation in orange juice at optimal conditions [44] |
| Nutritional Retention | Immediate increase in phenolics (5%) and anthocyanins (17%) in strawberry juice [44] | Slightly lower immediate enhancement but better long-term retention [44] |
| Color Preservation | Noticeable shifts in some juices; darker tones in orange juice [44] | Closest match to fresh juice appearance across juice types [44] |
| Processing Method | Continuous flow system [40] | Batch processing of pre-packaged products [44] |
| Operational Constraints | Requires electrical conductivity; ohmic heating effects [44] | High capital investment; packaging must withstand pressure [44] |
While both technologies effectively preserve nutrients compared to thermal processing, HPP generally demonstrates superior long-term microbial stability and color retention, whereas PEF offers advantages in continuous processing and potentially higher immediate nutrient enhancement [44].
Experimental Evidence and Methodologies
Representative Experimental Protocol for Liquid Food Preservation
Standardized methodologies have been developed to evaluate PEF efficacy in liquid food preservation. The following workflow illustrates a typical experimental approach for studying PEF processing of liquid foods like fruit juice.
A specific study on Thai orange juice exemplifies this approach, employing the following methodology [42]:
- Microbial Strains and Inoculation: Staphylococcus aureus TISTR 2329 and Escherichia coli TISTR 117 were inoculated into orange juice at initial concentrations of approximately 8.4 × 10⁵ CFU/mL and 8.9 × 10⁵ CFU/mL, respectively.
- PEF Treatment Parameters: Electric field strengths of 20, 30, and 40 kV/cm were applied with pulse numbers ranging from 10 to 50, pulse width of 10 μs, and frequency of 1 Hz, resulting in treatment times of 100-500 μs.
- Control Treatment: Conventional thermal pasteurization at 68.2°C for 30 minutes, following established protocols.
- Analysis Methods: Microbial viability was assessed using standard plate counting, cell morphology by scanning electron microscopy (SEM), and product quality through measurements of pH, color, viscosity, total soluble solids, vitamin C, sugars, and minerals.
This methodological approach provides comprehensive data on both microbial safety and quality retention, enabling direct comparison between PEF and conventional processing.
Key Research Reagent Solutions
The implementation of PEF research requires specific technical components and reagents. The table below details essential elements for conducting PEF experiments in liquid food preservation.
Table 3: Essential Research Reagents and Equipment for PEF Experiments
| Component | Specifications | Research Function |
|---|---|---|
| PEF Generator | High-voltage (20-80 kV/cm); Exponential decay or square wave pulses [40] | Generates controlled high-voltage pulses for electroporation |
| Treatment Chamber | Parallel plate or coaxial design; Electrode material (stainless steel 316L, carbon) [42] [40] | Houses sample during PEF application; electrode material affects corrosion resistance |
| Microbiological Media | Nutrient agar; Selective media for specific pathogens [42] | Cultivation and enumeration of microorganisms pre- and post-treatment |
| Analysis Reagents | HPLC standards for vitamins, sugars; Color measurement standards [42] | Quantification of nutritional and quality parameters |
| Buffer Solutions | Tris buffer (20 mM; pH 7.0) for enzyme studies [41] | Standardized media for studying enzyme inactivation kinetics |
| SEM Fixatives | Glutaraldehyde; Critical point drying equipment [42] | Sample preparation for microscopic examination of cell structural damage |
The selection of appropriate electrode materials is particularly important, as studies have noted that electrode constituents (e.g., Fe, Cr, Ni, Mn) may be released into food samples due to corrosion during PEF treatment. Carbon electrodes have been suggested to minimize this issue [40].
Research Gaps and Future Directions
Despite significant advances in PEF technology, several research gaps remain. While substantial literature exists on microbial inactivation by PEF, comparatively less information is available about its effects on food constituents and overall quality parameters during storage [39] [45]. More comprehensive studies are needed to understand the long-term stability of PEF-treated products and their bioactivity retention throughout shelf life.
The combination of PEF with other non-thermal technologies presents promising research avenues. Studies suggest synergistic effects when PEF is paired with methods such as ultraviolet radiation, high-intensity light pulses, ultrasound, or high-pressure carbon dioxide [40]. These hurdle approaches may enhance microbial inactivation while allowing reduced treatment intensities, potentially further improving quality retention.
Future research should also address the scaling challenges of PEF technology. While laboratory-scale results are promising, optimizing conditions for industrial-scale implementation requires additional investigation, particularly regarding electrode design, process uniformity, and energy efficiency [40]. The development of carbon electrodes and multiple treatment chambers in series represents progress in addressing field uniformity issues [40].
From a regulatory perspective, further research may help establish clearer guidelines for PEF processing. Although PEF is generally recognized as safe and has been used commercially for juice pasteurization in compliance with FDA regulations, continued studies on its effects on various food matrices will support broader regulatory acceptance and implementation [40].
Pulsed Electric Field technology represents a significant innovation in liquid food preservation, offering distinct advantages over conventional thermal processing methods, particularly in retaining heat-sensitive nutrients and fresh-like quality attributes. The principle of electroporation enables effective microbial inactivation while minimizing damage to food components, positioning PEF as a valuable technology for preserving bioactivity in processed foods.
Experimental evidence demonstrates that PEF treatment achieves comparable microbial safety to thermal pasteurization while superiorly preserving nutritional and sensory qualities. Although PEF shows some limitations in long-term microbial stability compared to High Pressure Processing, its continuous operation and energy efficiency make it an attractive alternative for specific applications.
For researchers and food industry professionals, PEF technology offers a promising approach to addressing consumer demands for minimally processed, nutritious foods while maintaining safety standards. Future developments in PEF equipment design, process optimization, and combination with other non-thermal technologies will likely expand its applications and effectiveness in preserving bioactive compounds in liquid food products.
In the field of food and pharmaceutical sciences, the extraction of bioactive compounds and surface decontamination have traditionally relied on thermal processing methods. However, the application of heat often leads to the degradation of thermolabile bioactive components, reducing the overall quality and efficacy of the final product [46] [2]. Non-thermal technologies, particularly cold plasma (CP) and ultrasound (US), have emerged as promising alternatives that effectively address these limitations while offering enhanced efficiency and sustainability [2].
Cold plasma, often described as the fourth state of matter, is a partially ionized gas generated at or near atmospheric pressure, containing a rich mixture of reactive oxygen and nitrogen species (RONS), electrons, ions, and ultraviolet photons [47]. Ultrasound utilizes high-frequency sound waves (typically 20-100 kHz) that generate cavitation bubbles in liquid media, producing intense local shear forces, turbulence, and mass transfer enhancements [46] [48]. When combined, these technologies create a powerful synergistic effect that maximizes extraction yields while preserving bioactive compounds and ensuring microbial safety, making them particularly valuable for research and development in nutraceutical and pharmaceutical applications [49] [50].
Fundamental Principles and Equipment
Table 1: Comparison of Cold Plasma and Ultrasound Technologies
| Parameter | Cold Plasma (CP) | Ultrasound (US) |
|---|---|---|
| Technology Type | Non-thermal plasma generated at atmospheric pressure | Acoustic cavitation via high-frequency sound waves |
| Primary Mechanisms | Reactive species (RONS) generation, UV radiation, ozone production | Cavitation, microjetting, shear forces, cell disruption |
| Key Equipment | Dielectric barrier discharge (DBD), plasma jet, corona discharge | Probe systems, ultrasonic baths |
| Typical Applications | Surface decontamination, pesticide degradation, extraction enhancement | Bioactive compound extraction, emulsion preparation, cell disruption |
| Treatment Scale | Surface-level and limited penetration | Bulk liquid penetration |
| Operational Parameters | Power (15-24 kV), exposure time (25-50 s), gas composition | Intensity (40-100%), time (1-90 min), temperature control |
Research Reagent Solutions and Essential Materials
Table 2: Essential Research Materials and Their Functions
| Material/Equipment | Function in Research Applications |
|---|---|
| Dielectric Barrier Discharge (DBD) System | Generates cold plasma for surface treatment and cell wall modification |
| Ultrasonic Probe System | Provides focused acoustic energy for cell disruption (typically 20-40 kHz) |
| Ultrasonic Bath | Offers uniform cavitation for smaller sample volumes |
| Natural Deep Eutectic Solvents (NADES) | Green extraction media for bioactive compounds |
| Fourier Transform Infrared (FTIR) Spectrometer | Analyzes molecular structure preservation in extracted compounds |
| DPPH/ABTS Reagents | Quantifies antioxidant activity in extracts |
| High-Performance Liquid Chromatography (HPLC) | Identifies and quantifies specific bioactive compounds |
| Response Surface Methodology (RSM) Software | Optimizes multiple extraction parameters efficiently |
Comparative Performance Analysis: Experimental Data
Extraction Efficiency and Bioactive Compound Recovery
Table 3: Quantitative Performance Comparison of Extraction Technologies
| Source Material | Technology | Optimal Conditions | Yield Improvement | Bioactive Enhancement | Reference |
|---|---|---|---|---|---|
| Rice bran | CP + US | CP: 20 min; US: 40% intensity, 40°C | 21.66% increased yield | 94.04% higher antioxidant activity | [49] |
| Spearmint leaves | CP + Ultrasonic bath | CP: 22.2 kV; UB: 40.1°C, 24.1 min | 155.7% higher EO yield | Increased oxygenated monoterpenes | [51] |
| Peach peels | CP + UAE | CAP pre-treatment + UAE: 17 min, 53% ethanol | Significant phenolic yield increase | Higher antioxidant activity vs. control | [50] |
| Caraway seeds | CP + US drying | CP: 25 s; US: 180 W, 39°C | 31% reduced drying time | 53.2% higher TPC, 43.6% higher TFC | [52] |
| Lentinula edodes | Ultrasound-assisted water | 20 kHz, 50-60% amplitude, 60°C max | Highest β-glucan content (34.51 g/100g) | - | [53] |
Synergistic Effects and Quality Preservation
The combination of cold plasma and ultrasound demonstrates significant synergistic effects that surpass their individual applications. In rice bran oil extraction, the combined approach not only increased yield but also reduced peroxide values and free fatty acids while better preserving tocopherols, tocotrienols, and the molecular structure of unsaturated fatty acids compared to solvent extraction alone [49]. FTIR and DSC analyses confirmed superior molecular structure preservation of heat-sensitive compounds when using the combined CP-US approach [49].
For essential oil extraction from spearmint, the CP-UB (ultrasonic bath) treatment increased oxygenated monoterpene content, particularly carvone, which is directly associated with enhanced antioxidant activity and overall oil quality [51]. This synergistic effect is attributed to CP's ability to disrupt surface structures and cellular membranes, followed by US-induced cavitation that facilitates the release of intracellular compounds into the extraction medium.
Experimental Protocols and Methodologies
Combined Cold Plasma and Ultrasound-Assisted Extraction
Figure 1: Experimental Workflow for CP-US Extraction
Sample Preparation Protocol
Plant materials should be dried at 25°C for approximately one week until stable, then ground to a consistent particle size (<0.5 mm) using a hammer mill [51]. The moisture content should be standardized, and samples stored at 4°C in sealed containers until processing [53].
Cold Plasma Pretreatment
Utilize a dielectric barrier discharge (DBD) system with the following parameters:
- Electrode configuration: Two parallel electrodes with dielectric barrier
- Power settings: 15-24 kV, 10 kHz pulse frequency, 300 W power
- Gas composition: Nitrogen, air, or argon at atmospheric pressure
- Exposure time: 25-50 seconds for seeds; 10-20 minutes for plant materials
- Sample placement: 3.5 mm distance between electrodes [52]
The pretreatment modifies cellular structures through the action of reactive oxygen and nitrogen species (RONS), creating micro-channels in cell walls that enhance solvent penetration in subsequent extraction steps [47].
Ultrasound-Assisted Extraction
Immediately following CP pretreatment, subject samples to UAE with optimized parameters:
- Equipment: Probe-type ultrasonic system (20-40 kHz frequency)
- Intensity: 40-60% amplitude (approximately 750 W maximum power)
- Temperature: 40-60°C (controlled with external water bath)
- Duration: 10-90 minutes in intermittent mode (4s on, 3s off)
- Solvent system: Ethanol:water (50-70% v/v) or NADES
- Solvent-to-solid ratio: 30:1 to 70:1 mL/g [49] [50]
The cavitation effects disrupt cell walls and enhance mass transfer, facilitating the release of intracellular compounds into the solvent medium [48].
Analytical Methods for Quality Assessment
Bioactive Compound Quantification
- Total Phenolic Content (TPC): Folin-Ciocalteu method, expressed as mg gallic acid equivalents (GAE)/g extract [50]
- Total Flavonoid Content (TFC): Aluminum chloride colorimetric method [52]
- Antioxidant Activity: DPPH and ABTS radical scavenging assays, FRAP assay [49] [52]
- Specific Bioactives: HPLC analysis for quantification of individual compounds (e.g., chlorogenic acid, carvone, β-glucan) [51] [53]
Structural and Quality Analysis
- FTIR Spectroscopy: Molecular structure preservation of unsaturated fatty acids and triglycerides [49]
- DSC (Differential Scanning Calorimetry): Thermal behavior and oxidative stability [49]
- Peroxide Value and Free Fatty Acids: Indicators of oxidative degradation [49]
- Color Measurement: Colorimeter for total color change (ΔE) [52]
Mechanisms of Action: Scientific Foundations
Synergistic Enhancement Mechanisms
Figure 2: Mechanism of CP-US Synergistic Action
The enhanced efficiency of combined cold plasma and ultrasound treatment stems from complementary mechanisms acting at different structural levels. Cold plasma primarily functions through the generation of reactive oxygen and nitrogen species (RONS), including ozone, hydroxyl radicals, and atomic oxygen, which progressively etch and erode the surface structures of biological materials [47]. This creates micro-channels and increases surface area for improved solvent penetration. Simultaneously, UV radiation from plasma contributes to microbial inactivation and can modify structural polymers [47].
Ultrasound complements this action through acoustic cavitation, where the formation, growth, and implosive collapse of microscopic bubbles generates intense local shear forces, microjetting, and turbulence [48]. These physical effects further disrupt the plasma-weakened cellular structures, facilitating the release of intracellular compounds. The combined approach also enhances mass transfer rates by reducing particle size and increasing diffusion coefficients, allowing for more efficient compound recovery at lower temperatures compared to conventional methods [49] [48].
The comparative analysis demonstrates that cold plasma and ultrasound technologies offer significant advantages over thermal processing methods for applications requiring bioactive compound preservation and surface decontamination. The synergistic combination of these non-thermal technologies enables higher extraction yields, reduced processing times, enhanced energy efficiency, and superior preservation of thermolabile bioactive compounds [49] [51] [50].
For researchers and drug development professionals, these technologies present opportunities to develop more efficient and sustainable extraction protocols for nutraceutical and pharmaceutical applications. The ability to customize treatment parameters (power, duration, intensity) for specific biological materials provides flexibility in process optimization [52]. Future research directions should focus on scaling up these technologies for industrial applications, optimizing sequential versus simultaneous treatment approaches, and further exploring the mechanisms underlying their synergistic effects across different biological matrices.
Non-thermal processing technologies have emerged as promising alternatives to conventional thermal methods, offering effective microbial control while minimizing damage to heat-sensitive bioactive compounds. Among these, ultraviolet (UV) and Pulsed Light (PL) technologies represent distinct, chemical-free approaches to surface disinfection and shelf-life extension. This guide provides an objective comparison of these technologies, focusing on their performance characteristics, experimental applications, and effects on food quality and bioactivity within the broader context of thermal versus non-thermal processing research.
UV light for food processing primarily uses UV-C wavelengths (200–280 nm), which are absorbed by microbial DNA, causing lethal damage and preventing replication [54] [55]. PL is a broader-spectrum technology that uses short, high-intensity pulses of light, rich in UV-C, but also containing visible and infrared wavelengths (200–1100 nm) [56]. The U.S. Food and Drug Administration (FDA) has approved both technologies for food production, processing, and handling [54] [56].
Technology Comparison: Fundamental Principles and Characteristics
The following table summarizes the core characteristics of UV and Pulsed Light technologies.
Table 1: Fundamental characteristics of UV and Pulsed Light technologies
| Characteristic | Ultraviolet (UV) Light | Pulsed Light (PL) |
|---|---|---|
| Primary Mechanism | Primarily photochemical (DNA damage) [55] | Combined photochemical, photothermal, and photophysical effects [56] |
| Spectral Range | Narrow spectrum, typically 200–280 nm (UV-C) [54] | Broad spectrum: UV (200–400 nm), Visible (400–700 nm), IR (700–1100 nm) [56] |
| Treatment Nature | Continuous wave or low-intensity [55] | Pulsed, high-intensity, short duration (μs to ms) [57] [56] |
| FDA Limit | Not specified for continuous UV | Cumulative treatment ≤ 12 J/cm²; pulse width ≤ 2 ms [56] |
| Penetration Depth | Low, surfaces only [54] [57] | Low, surfaces only [57] |
| Typical Applications | Surface decontamination of foods, equipment, water, and air [54] [58] | Surface decontamination of foods, packaging materials, and equipment [57] [56] |
Comparative Performance Data: Efficacy and Quality Impacts
Microbial Inactivation Efficacy
Both technologies achieve significant microbial log reductions, though their efficacy depends on treatment parameters and target organisms.
Table 2: Comparison of microbial inactivation efficacy
| Technology | Test Material / Surface | Conditions / Parameters | Microbial Reduction | Key Findings |
|---|---|---|---|---|
| UV Light | Chilled beef [54] | 254 nm, 6 cm distance, 14 s | 0.8 log CFU/g initial reduction | Total bacterial count reduced by 0.56–1.51 log CFU/g during storage. |
| UV Light | Ground beef [54] | 254 nm, 800 μW/cm², 30 s | ~1.0 log CFU/g | Improvement in Salmonella control. |
| UV Light | Donkey milk whey protein [8] | Non-thermal treatment | N/A | Preserved lysozyme, lactoferrin, and IgG activities better than heat. |
| Pulsed Light | In vitro (agar plates) [56] | 1–20 flashes, 0.01–50 J/cm² | Significant reduction | Rapid inactivation of Listeria monocytogenes within seconds. |
| Pulsed Light | Food packaging materials [57] | Broad spectrum, pulsed | Varies | Efficacy depends on surface topography, reflectivity, and microbial type. Gram-negative bacteria are generally most susceptible. |
Impact on Product Quality and Bioactivity
A critical advantage of non-thermal technologies is their potential to better preserve nutritional and sensory qualities compared to thermal processing.
Table 3: Impact on food quality and bioactive components
| Technology | Product | Effect on Quality & Bioactivity | Comparison to Thermal Processing |
|---|---|---|---|
| UV Light | Chilled beef [54] | No adverse impact on pH, color, or sensory quality. Slight increase in TBARS (lipid oxidation) during late storage. | Thermal processing often leads to greater nutrient loss and sensory degradation [2]. |
| UV Light | Fruits & Vegetables [7] | Can induce accumulation of phytochemicals like ascorbic acid and phenolic compounds. | Unlike thermal treatments, which reduce bioactive compounds, UV can enhance them. |
| UV Light | Donkey milk whey [8] | Oxidized internal disulfide bonds, increasing free sulfhydryl groups; improved foaming/emulsification stability. | High-temperature treatments caused severe damage; lysozyme activity preservation was only 23% with heat vs. much higher with UV. |
| Pulsed Light | Various Foods [56] | Minimal impact on sensory and nutritional quality at optimal doses. Possible temperature rise (IR component) may affect organoleptic properties. | Leaves no residues, unlike some chemical disinfectants used in packaging sterilization [57]. |
Experimental Protocols and Methodologies
Detailed Protocol: UV Treatment for Meat Shelf-life Extension
The following methodology, derived from a study on chilled beef, can serve as a template for evaluating UV efficacy [54].
- Sample Preparation: Obtain uniform portions of the target food (e.g., 250 g beef longissimus dorsi). Trim visible connective and adipose tissue. Package samples individually in sterile, UV-transparent packaging material (e.g., polyethylene bags).
- UV Treatment Setup: Use a UV-C lamp emitting at 254 nm. Ensure the UV intensity is calibrated using a ultraviolet radiation meter at the treatment distance.
- Optimization Phase: Conduct a factorial experiment with varying irradiation distances (e.g., 6, 9, 12 cm) and irradiation times (e.g., 6, 10, 14 s). Include a non-treated control group.
- Evaluation Metrics: Analyze treated and control samples for:
- Microbial Load: Total bacterial count (log CFU/g).
- Chemical Quality: Total volatile basic nitrogen (TVB-N), Thiobarbituric acid reactive substances (TBARS) for lipid oxidation, and pH.
- Physical/Sensory Quality: Color (e.g., using a colorimeter), and sensory evaluation.
- Storage Study: Apply the optimal treatment parameters (e.g., 6 cm for 14 s from the cited study) to a new batch. Monitor the above metrics periodically during refrigerated storage (e.g., 0–15 days at 0°C) and compare against a control.
UV Treatment Experimental Workflow
Detailed Protocol: Pulsed Light for Surface Decontamination
This protocol is adapted from research on sterilizing food packaging and contact surfaces [57] [56].
- Surface Inoculation: Prepare a microbial culture (e.g., E. coli, S. aureus, L. innocua) in a suitable broth. For solid surfaces, spot-inoculate or spray with a uniform microbial suspension and air-dry under laminar flow.
- PL Equipment Setup: Use a PL system with a flashlamp filled with inert gas (e.g., Xenon). The system should emit broad-spectrum light (200–1100 nm). Set the pulse width (e.g., ≤ 2 ms) and number of pulses based on the target fluence (J/cm²).
- Treatment Application: Place inoculated samples at a fixed distance from the light source. Treat samples with a predetermined fluence (e.g., from 0.01 to 5 J/cm², ensuring the cumulative treatment does not exceed 12 J/cm² as per FDA).
- Microbial Enumeration: After treatment, transfer the surface (or a defined area) into a neutralizing solution and vortex to recover microorganisms. Perform serial dilutions and plate on appropriate agar media. Incubate and count colonies to calculate log reduction compared to a non-treated control.
- Surface Analysis: (Optional) Use techniques like scanning electron microscopy (SEM) to observe physical damage to microbial cells or Fourier transform infrared spectroscopy (FTIR) to analyze chemical changes on the treated surface.
Mechanisms of Microbial Inactivation
The germicidal action of UV and PL involves distinct but sometimes overlapping pathways.
Microbial Inactivation Pathways
- UV Light Mechanism: The primary mechanism is photochemical. The UV-C radiation, particularly at 253.7 nm, is absorbed by the microbial DNA, leading to the formation of cyclobutene-pyrimidine dimers (CPDs) and pyrimidine (6-4) pyrimidone photoproducts (6-4PP). These lesions inhibit transcription and replication, leading to cell death [54] [55].
- Pulsed Light Mechanisms: PL employs a multi-mechanism approach [55] [56]:
- Photochemical: The UV component acts similarly to continuous UV light.
- Photothermal: The intense, short-duration pulses, particularly from the visible and IR spectra, can cause rapid localized heating of microbial cells, leading to protein denaturation and membrane rupture.
- Photophysical: The high peak power of the pulses can physically disrupt cellular structures, causing cell wall damage, cytoplasmic membrane shrinkage, and leakage of cellular contents.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key reagents and materials for UV and PL research
| Item | Function / Application | Example / Specification |
|---|---|---|
| UV-C Lamp | Source of continuous ultraviolet light. | Low-pressure mercury lamp, 254 nm wavelength [54]. |
| Pulsed Light System | Source of high-intensity, broad-spectrum pulsed light. | Xenon flashlamp system; spectrum 200-1100 nm; pulse width ≤ 2 ms [56]. |
| Ultraviolet Radiation Meter | Measuring UV light intensity at the sample surface. | Calibrated meter for 254 nm (e.g., UV-254) [54]. |
| Microbiological Media | Culturing and enumerating microorganisms pre- and post-treatment. | Tryptic Soy Agar (TSA), Plate Count Agar (PCA), selective media for pathogens [54]. |
| Neutralizing Solution | Halting antimicrobial activity after treatment for accurate microbial recovery. | D/E Neutralizing Broth or Buffered Peptone Water [56]. |
| UV-Transparent Packaging | Holding samples during treatment without blocking UV light. | Polyethylene bags (e.g., 0.025 mm thickness) [54]. |
| Colorimeter | Quantifying color changes in food samples post-treatment. | CIE L, a, b* color space measurement [54]. |
| Chemical Assay Kits | Quantifying chemical quality parameters. | TBARS (for lipid oxidation), TVB-N (for protein degradation) [54]. |
UV and Pulsed Light technologies offer effective non-thermal solutions for surface disinfection and shelf-life extension. UV provides a targeted, primarily photochemical approach suitable for continuous decontamination processes. In contrast, Pulsed Light delivers a high-intensity, multi-mechanism "kill" in short bursts, ideal for rapid treatment of surfaces and packaging. The choice between them depends on the specific application, required log reduction, sensitivity of the product, and processing constraints. Crucially, both can achieve microbial safety while better preserving the bioactive compounds and sensory attributes of products compared to traditional thermal processing, aligning with the growing demand for high-quality, minimally processed foods. Future research will likely focus on optimizing parameters for diverse products, overcoming shadowing effects, and integrating these technologies with other non-thermal methods in hurdle approaches for enhanced efficacy.
Overcoming Practical Hurdles: Scaling, Optimization, and Economic Viability
Addressing Scalability and High Initial Investment Costs
The transition from conventional thermal to non-thermal processing technologies represents a significant paradigm shift in food and pharmaceutical research, driven by the need to better preserve heat-sensitive bioactive compounds. While thermal technologies like pasteurization and sterilization effectively ensure microbial safety, they often degrade nutritional quality, causing undesirable biochemical changes, loss of vitamins, and reduction of bioactive phytochemicals [59]. Non-thermal technologies—including High-Pressure Processing (HPP), Pulsed Electric Fields (PEF), Cold Plasma (CP), and Ultraviolet Light (UV)—have emerged as promising alternatives that effectively inactivate microorganisms and enzymes while operating at ambient or mild temperatures, thereby minimizing thermal degradation [22] [60].
However, the adoption of these innovative technologies faces two significant interconnected barriers: scalability challenges and high initial investment costs. For researchers and development professionals evaluating these technologies for bioactivity research, understanding the economic and operational feasibility at various scales is crucial. This guide provides a systematic comparison of these technologies, focusing on their scalability, economic viability, and experimental implementation to inform strategic decision-making in research and development contexts.
Technology Comparison: Scalability and Economic Parameters
Comparative Analysis of Key Non-Thermal Technologies
Table 1: Scalability and Investment Comparison of Non-Thermal Processing Technologies
| Technology | Typical Scale-Up Readiness (TRL) | Initial Investment Cost | Key Scalability Constraints | Energy Consumption | Footprint & Integration |
|---|---|---|---|---|---|
| High-Pressure Processing (HPP) | Commercial (High TRL) | Very High [61] | Batch processing limits throughput; vessel pressure capacity | High during compression phase [61] | Significant space requirements; batch system |
| Pulsed Electric Field (PEF) | Pilot to Commercial (Medium-High TRL) | High [62] | Chamber design for uniform field distribution; flow rate optimization | Improved efficiency with heat recovery [62] | Continuous flow; easier line integration |
| Cold Plasma (CP) | Lab to Pilot (Low-Medium TRL) | Moderate to High [61] | Surface treatment limitation; gas composition control at scale | Highly energy efficient [2] | Various configurations possible |
| Ultraviolet (UV) | Commercial (High TRL) | Lower relative cost [61] | Penetration depth in opaque liquids; fouling of lamps | Low energy requirements [2] | Compact systems; easy integration |
Quantitative Economic Analysis
Table 2: Cost Structure and Environmental Impact Comparison (Case Study: Orange Juice Processing)
| Processing Technology | Relative Selling Price | Carbon Footprint | Energy Reduction Potential | Payback Period Considerations |
|---|---|---|---|---|
| Thermal Pasteurization | Baseline | Baseline | Baseline | Baseline |
| HPP | Highest [61] | Comparable or lower [61] | Moderate | Lengthy due to high capex |
| PEF | Slightly higher [61] | ~30% lower GHG emissions [62] | 20% electricity, >60% fuel gas reduction [62] | Improved with heat recovery integration |
| UV | Competitive | Lower | Significant | Most favorable |
Experimental Protocols for Technology Evaluation
Standardized Bioactivity Preservation Assessment
Objective: Quantify the retention of heat-sensitive bioactive compounds (e.g., vitamins, polyphenols, carotenoids) following non-thermal processing compared to conventional thermal treatment.
Methodology:
- Sample Preparation: Prepare uniform homogenates of plant-based materials (e.g., fruit/vegetable juices) to ensure consistent matrix composition
- Processing Conditions:
- Bioactivity Analysis:
- Total Phenolic Content: Folin-Ciocalteu method
- Antioxidant Capacity: ORAC and DPPH assays
- Vitamin C Retention: HPLC quantification
- Carotenoid Profile: HPLC-DAD analysis
- Microbial Validation: Ensure 5-log reduction of pertinent microorganisms per FDA guidelines [60]
Scalability Assessment Protocol
Objective: Evaluate the feasibility of scaling non-thermal processes from laboratory to pilot or industrial scale.
Methodology:
- Throughput Analysis: Conduct capacity scaling experiments at 20 L/h (lab), 200 L/h (pilot), and 2000 L/h (industrial) for continuous flow systems like PEF [62]
- Energy Efficiency Monitoring: Measure specific energy input (kJ/kg) across scales and compare with microbial inactivation efficacy
- Quality Consistency Testing: Assess bioactive compound retention variability across processing batches and scales
- Process Integration Simulation: Evaluate compatibility with existing thermal equipment for hybrid approaches
Technology Selection Framework
Research Reagent Solutions for Experimental Implementation
Table 3: Essential Research Materials for Non-Thermal Processing Studies
| Reagent/Material | Function in Experimental Research | Application Examples |
|---|---|---|
| Folin-Ciocalteu Reagent | Quantification of total phenolic content via colorimetric assay | Measuring polyphenol retention in plant extracts [63] |
| DPPH (2,2-diphenyl-1-picrylhydrazyl) | Assessment of free radical scavenging capacity | Antioxidant activity evaluation in processed juices [63] |
| HPLC Standards (ascorbic acid, carotenoids, phenolic compounds) | Calibration for quantitative analysis of specific bioactive compounds | Vitamin C and carotenoid quantification [63] |
| Selective Media (PCA, PDA, MEA) | Microbial enumeration for safety and efficacy validation | 5-log reduction verification per FDA guidelines [60] |
| Pressure-Transmitting Fluid | Medium for uniform pressure distribution in HPP | Food-grade propylene glycol-water mixtures [61] |
| Conductivity Adjustment Salts | Optimization of electrical conductivity for PEF treatment | Potassium chloride or sodium chloride solutions [62] |
Non-thermal processing technologies present a compelling alternative to conventional thermal methods for preserving bioactive compounds in food and pharmaceutical applications. The superior bioactivity retention demonstrated by technologies like HPP and PEF must be balanced against their significant economic considerations, particularly regarding scalability and initial investment.
For research and development applications, the selection framework presented enables systematic evaluation based on specific project requirements. PEF technology emerges as a particularly promising option, offering a favorable balance between bioactivity preservation, energy efficiency, and scalability, especially when integrated with heat recovery systems. HPP delivers exceptional quality retention but faces constraints in continuous processing applications. UV and cold plasma technologies offer more accessible entry points for research programs with budget limitations, though with certain application constraints.
Future development should focus on optimizing hybrid approaches that combine the strengths of multiple technologies while addressing scalability challenges through equipment modularization and process intensification. As these technologies mature and adoption increases, initial investment costs are expected to decrease, making them more accessible for broader research and commercial applications.
In the field of food and bioactive component processing, the choice between thermal and non-thermal technologies represents a critical crossroads with significant implications for final product quality, functionality, and bioactivity. While thermal processing remains a conventional and effective method for microbial inactivation, its application often comes at the expense of heat-sensitive bioactive compounds and nutritional quality [22] [5]. In response to growing consumer demand for fresh-like, minimally processed foods with retained health benefits, non-thermal technologies have emerged as promising alternatives that operate at or near ambient temperatures [60] [64]. This comparative guide objectively examines the critical processing parameters—pressure, time, intensity, and temperature—across both thermal and non-thermal technologies, with particular focus on their effects on bioactive preservation, providing researchers and scientists with experimental data and methodologies to inform process optimization.
Comparative Analysis of Processing Parameters and Bioactive Retention
The fundamental distinction between thermal and non-thermal technologies lies in their primary mechanism of microbial inactivation: thermal processing relies on heat energy, while non-thermal methods utilize alternative physical principles such as pressure, electric fields, or electromagnetic radiation, resulting in markedly different impacts on bioactive compounds [65] [5].
Table 1: Critical Parameter Ranges and Bioactive Impact of Thermal vs. Non-Thermal Technologies
| Processing Technology | Pressure/Intensity Parameters | Time Parameters | Temperature Parameters | Impact on Bioactive Compounds |
|---|---|---|---|---|
| Thermal Processing | N/A | Seconds to minutes | 63-121°C (pasteurization to sterilization) | Significant degradation of heat-sensitive vitamins, polyphenols, and carotenoids; protein denaturation [7] [8] |
| High-Pressure Processing (HPP) | 100-900 MPa | 1.5-15 minutes | Ambient to 45°C (for pasteurization) | Excellent retention of low-molecular-weight compounds (vitamins, pigments, flavor compounds); minimal effect on covalent bonds [66] [60] |
| Pulsed Electric Field (PEF) | 20-80 kV/cm | Microseconds to milliseconds | <50°C (can be non-thermal) | Better retention of heat-sensitive compounds compared to thermal processing; may enhance extraction of bioactives [60] [64] |
| Ultrasound Treatment | 20-100 kHz (frequency) | 1-60 minutes | Slight increase due to cavitation | Can enhance extraction of bioactives; potential oxidative degradation at high intensities [5] [64] |
| Cold Plasma | Varies with system design | Seconds to minutes | 30-60°C | Surface-dependent effects; can induce bioactive formation in some whole fruits and vegetables [22] [7] |
| UV Light | Varies with wavelength and intensity | Seconds to minutes | Ambient temperature | Surface treatment only; can induce phytochemical accumulation in some produce [7] |
Table 2: Direct Experimental Comparison of Thermal vs. Non-Thermal Effects on Bioactive Components
| Study Reference | Processing Technology | Critical Parameters | Key Findings on Bioactive Components |
|---|---|---|---|
| Donkey milk whey protein study [8] | High-Temperature Long-Time (HTLT) thermal | High temperature for extended time | Lysozyme activity preservation: only 23%; Lactoferrin: 70% loss; IgG: 77% loss |
| Donkey milk whey protein study [8] | Ultrasonication (non-thermal) | Specific frequency and time (methodology detailed in Section 3) | Notable reduction in protein particle size and turbidity; better preservation of active components compared to thermal |
| Donkey milk whey protein study [8] | UV treatment (non-thermal) | Specific intensity and time (methodology detailed in Section 3) | Oxidation of internal disulfide bonds, increasing free sulfhydryl groups; improved foaming and emulsification stability |
| Fruit juice processing [60] | Thermo-sonication with nisin | Combination of mild heat and ultrasound with antimicrobial | Increased total polyphenols (10.03%), total carotenoids (20.10%), and antioxidant capacity (51.10% by ORAC) |
| Carrot-based products [7] | Pulsed Electric Fields (PEF) | 5 pulses of 3.5 kV·cm−1; 0.6 kJ·kg−1 | Phenolic bio-accessibility reached 100% in purees; thermally treated oil-added purees showed greatest carotenoid bio-accessibility (7.8%) |
Experimental Protocols for Comparative Analysis
Protocol 1: Comparative Analysis of Thermal vs. Non-Thermal Treatments on Whey Protein Structure and Functionality
This methodology is adapted from the experimental approach used in the donkey milk whey protein study [8].
Objective: To evaluate and compare the effects of thermal and non-thermal processing methods on the structural and functional properties of whey proteins, with specific attention to preservation of bioactive components.
Materials and Reagents:
- Raw donkey milk (or other protein source of interest)
- High-temperature long-time (HTLT) thermal processing equipment
- Ultrasonication apparatus (e.g., ultrasonic probe system, 20-100 kHz frequency range)
- UV treatment system (appropriate wavelength and intensity for food applications)
- Fluorometer for measuring fluorescence intensity
- Spectrophotometer for measuring turbidity
- Enzyme activity assays for lysozyme, lactoferrin, and IgG
- Equipment for foaming and emulsification stability tests
Methodology:
- Sample Preparation: Separate whey proteins from raw milk using standard centrifugation and filtration techniques.
- Thermal Treatment: Subject aliquots of whey protein solution to HTLT conditions (e.g., 85-90°C for 15-30 minutes).
- Ultrasonication Treatment: Treat whey protein aliquots using an ultrasonic probe at controlled frequency (e.g., 20 kHz) and duration (e.g., 5-15 minutes), with temperature control to prevent thermal effects.
- UV Treatment: Expose whey protein aliquots to UV light at controlled intensity and duration, ensuring even exposure.
- Analysis:
- Measure structural changes via fluorescence intensity and turbidity
- Quantify preservation of bioactive components (lysozyme, lactoferrin, IgG) using specific activity assays
- Evaluate functional properties through foaming and emulsification stability tests
Protocol 2: Enhanced Bioactive Compound Extraction and Retention in Plant-Based Matrices
This methodology integrates approaches from multiple studies on fruit and vegetable processing [7] [5].
Objective: To compare the efficacy of thermal and non-thermal technologies in extracting and retaining bioactive compounds from plant matrices, and to evaluate the bio-accessibility of these compounds.
Materials and Reagents:
- Fresh plant material (e.g., carrots, fruits, or vegetables)
- Pulsed Electric Field (PEF) equipment (capable of 3-4 kV·cm−1)
- Thermal processing equipment (water bath or heat exchanger)
- High-Pressure Processing equipment (if available)
- Analytical equipment for polyphenol, carotenoid, and antioxidant capacity analysis (HPLC, spectrophotometer)
- In vitro digestion model components
Methodology:
- Sample Preparation: Prepare plant materials according to target products (juices, purees, or oil-added purees).
- PEF Treatment: Apply PEF treatment at optimized parameters (e.g., 3.5 kV·cm−1, 5 pulses) to plant material before product formation.
- Thermal Treatment: Apply conventional thermal pasteurization conditions (e.g., 72-85°C for 15-30 seconds) to comparable samples.
- Product Formation: Process treated materials into final products (juices, purees, etc.).
- Analysis:
- Quantify bioactive compounds (polyphenols, carotenoids) pre- and post-processing
- Measure antioxidant capacity using multiple assays (ORAC, DPPH)
- Evaluate bio-accessibility using in vitro digestion models
- Assess potential degradation products (e.g., hydroxymethyl-L-furfural in thermal treatments)
Visualizing Processing Pathways and Effects
The following diagrams illustrate the fundamental mechanisms of thermal versus non-thermal processing technologies and their distinct effects on food components and bioactive compounds.
Figure 1: Fundamental Mechanisms of Thermal vs. Non-Thermal Processing Technologies
Figure 2: Parameter Optimization Workflow for Thermal and Non-Thermal Processing
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Equipment for Processing and Analysis
| Item Category | Specific Examples | Function in Research |
|---|---|---|
| Processing Equipment | High-Pressure Processing (HPP) pilot-scale unit | Applies isostatic pressures (100-900 MPa) for microbial inactivation and bioactive retention studies |
| Pulsed Electric Field (PEF) system | Generates high-voltage pulses (20-80 kV/cm) for cell membrane disruption studies | |
| Ultrasonication apparatus (probe or bath) | Generates sound waves (>20 kHz) for extraction enhancement and microbial inactivation studies | |
| Precision thermal processing equipment | Provides controlled thermal treatments for comparative studies | |
| Analytical Reagents & Kits | Enzyme activity assays (lysozyme, lactoferrin, IgG) | Quantifies preservation of bioactive proteins after processing |
| Antioxidant capacity assay kits (ORAC, DPPH) | Measures retention of antioxidant properties post-processing | |
| Polyphenol and carotenoid quantification standards | Enables precise measurement of specific bioactive compounds | |
| Protein structure analysis reagents (e.g., for SH group quantification) | Evaluates structural changes in proteins after processing | |
| Cell Culture & Microbiology | Microbial strains (Listeria, Salmonella, E. coli) | Validation of microbial inactivation efficacy of processing parameters |
| Culture media and enumeration supplies | Quantifies log reductions achieved by different processing conditions | |
| Spore-forming bacterial strains | Studies on spore inactivation in combined thermal-pressure processes |
The optimization of critical parameters—pressure, time, intensity, and temperature—in both thermal and non-thermal processing technologies requires careful consideration of the target outcomes, particularly when bioactive preservation is a primary objective. Thermal processing, while effective for microbial inactivation, demonstrates significant limitations in preserving heat-sensitive bioactive compounds, as evidenced by the substantial losses of lysozyme, lactoferrin, and IgG in whey proteins [8]. Non-thermal technologies offer compelling alternatives with superior bioactive retention profiles, though they present their own optimization challenges and parameter dependencies. The choice between these technological pathways must be guided by the specific bioactive components of interest, the desired functional properties in the final product, and the required microbial safety outcomes. Future research directions should focus on optimizing combination processes that leverage the microbial inactivation efficacy of thermal methods with the bioactive preservation capabilities of non-thermal technologies, ultimately enabling the production of safer, more nutritious food products with enhanced health-promoting properties.
Combining Non-Thermal Methods for Enhanced Efficacy and Efficiency
In the field of food processing and pharmaceutical development, the preservation of bioactive compounds is paramount. Conventional thermal processing has been the cornerstone for microbial inactivation and shelf-life extension for decades. However, its major drawback lies in the degradation of heat-sensitive nutrients and bioactives; for instance, boiling carrots leads to a significant loss of phenolic compounds and vitamins [67]. This degradation directly impacts the efficacy of products derived from these processes, a critical concern for researchers and drug development professionals seeking to maximize the therapeutic potential of bioactive compounds. In response, non-thermal processing technologies have emerged as a superior alternative for preserving nutritional and functional properties while ensuring safety.
These non-thermal methods, which include High-Pressure Processing (HPP), Pulsed Electric Fields (PEF), Cold Plasma (CP), and Ultraviolet (UV) light, inactivate microorganisms through mechanisms other than heat, such as pressure-induced cell membrane damage or electrical breakdown [17]. While effective as standalone treatments, a more sophisticated approach is gaining traction: the combination of non-thermal methods. This synergistic strategy, often integrated with hurdle technology, aims to enhance efficacy and efficiency, potentially overcoming the limitations of single-method applications and offering researchers a more powerful toolkit for processing heat-labile bioactive substances [17].
Comparative Efficacy: Quantitative Data on Processing Methods
A direct comparison of the environmental, economic, and qualitative impacts of different processing methods is essential for informed decision-making in research and development.
Table 1: Environmental Impact and Cost Analysis of Non-Thermal vs. Thermal Processing
| Processing Technology | Carbon Footprint | Operational Cost (Example: Orange Juice) | Energy Consumption |
|---|---|---|---|
| Thermal Pasteurization | Baseline | ~1.5 US¢/L [68] | High, energy-intensive due to sustained heating [43] |
| High-Pressure Processing (HPP) | Comparable or lower than thermal [61] | ~10.7 US¢/L [68] | Concentrated in compression phase; no energy to maintain pressure [61] |
| Pulsed Electric Field (PEF) | Lower carbon footprint [43] | Information Missing | High efficiency; up to 50% less energy than thermal methods [43] |
| Cold Plasma (CP) | Information Missing | Information Missing | Information Missing |
| Ultraviolet (UV) Light | Information Missing | Information Missing | Information Missing |
Table 2: Impact on Bioactive Compounds and Food Quality
| Processing Method | Impact on Carotenoids | Impact on Phenolic Compounds & Antioxidants | Impact on Vitamins (e.g., C, B9, B12) |
|---|---|---|---|
| Boiling (Thermal) | Increase (e.g., in carrots) [67] | Significant loss [67] | Significant decline [67] |
| Steaming (Thermal) | Information Missing | Preserves antioxidant properties better than boiling [67] | Information Missing |
| High-Pressure Processing (HPP) | Information Missing | Maintains fresh-like attributes and bioactive compounds [61] | Preserves heat-sensitive vitamins [68] |
| Pulsed Electric Field (PEF) | Information Missing | Preserves antioxidants and flavors [43] | Preserves vitamins and enzymes [43] |
| Cold Plasma (CP) | Information Missing | Can preserve and enhance release of bioactive compounds in cereal bran [69] | Information Missing |
Synergistic Combinations of Non-Thermal Methods
The integration of multiple non-thermal technologies can induce more pronounced structural changes in biological matrices than single treatments alone, leading to significantly enhanced efficacy [69]. This synergistic approach is particularly promising for unlocking hard-to-extract bioactive compounds or achieving microbial inactivation levels comparable to thermal methods without the damaging heat effects.
A prime example of this synergy is the combined application of PEF and HPP. Research has demonstrated that this combination can achieve a significant reduction of 14-29% in aflatoxin levels in grape juice, with individual aflatoxin reductions as high as 84% [17]. This is a clear enhancement over the capabilities of either technology used independently. Similarly, in the processing of cereal bran, which is rich in dietary fiber and polyphenols, combined non-thermal methods have proven superior. The rigid cell wall structure of bran limits the release of its bioactive compounds. While single non-thermal methods can help, combined applications induce more pronounced structural changes, resulting in significantly higher release levels of bioactive compounds and enhanced antioxidant and nutritional functionality [69].
The following workflow outlines a generalized experimental approach for applying combined non-thermal methods to a biological material to enhance bioactivity:
Detailed Experimental Protocols for Key Studies
To ensure reproducibility and provide a clear technical foundation, this section details the methodologies from key studies cited in this guide.
Protocol for HPP and PEF Combination on Aflatoxin Reduction
This protocol is derived from a study investigating the synergistic effect of HPP and PEF in reducing aflatoxins in grape juice [17].
- 1. Sample Preparation: Obtain fresh grape juice. Standardize the initial microbial load and aflatoxin concentration (e.g., aflatoxin B1, B2, G1, G2) through pre-treatment analysis.
- 2. PEF Treatment:
- Equipment: A PEF system with a treatment chamber and a high-voltage pulse generator.
- Parameters: Apply a field strength of 2.7 kV/cm with a pulse width ranging from 15 to 1000 μs [17]. Ensure the juice is flowing continuously through the chamber during treatment.
- 3. HPP Treatment:
- Equipment: An HPP unit with a pressure vessel capable of reaching at least 600 MPa.
- Parameters: Subject the PEF-treated juice to a pressure range of 200-600 MPa. The holding time should be between 2 to 5 minutes at ambient or chilled temperature [61].
- 4. Analysis:
- Aflatoxin Quantification: Use high-performance liquid chromatography (HPLC) with fluorescence detection to quantify the concentration of individual aflatoxins post-treatment. Calculate the percentage reduction for each aflatoxin.
- Microbial Analysis: Plate count methods can be used to determine the reduction in total microbial load, if applicable.
Protocol for Non-Thermal Sterilization of Pulse Flours for Fermentation
This protocol, adapted from Paff & Cockburn (2023), describes the use of liquid chemical sterilants as non-thermal alternatives to autoclaving heat-labile materials like pulse flours for in vitro fermentation experiments [70].
- 1. Sample Preparation: Use flours from green lentil, field pea, chickpea, or similar. The flour should be raw and not pre-treated with heat.
- 2. Chemical Treatment (choose one):
- Bleach Treatment: Dilute germicidal bleach (8.25% sodium hypochlorite) with deionized water to a final concentration of 1.0% or 1.5% sodium hypochlorite. Mix the flour with the bleach solution for a specified contact time.
- Hydrogen Peroxide Treatment: Dilute 30% hydrogen peroxide with deionized water to a final concentration of 2.0%. Mix the flour with the hydrogen peroxide solution.
- Alcohol Treatment (Less Effective): Dilute 90% reagent alcohol with deionized water to a final concentration of 70% ethanol. Mix the flour with the alcohol solution. Note: This method was found to be less effective, with detectable microbes remaining after treatment [70].
- 3. Residual Chemical Removal: After treatment, remove the residual liquid sterilant from the insoluble flour substrate using methods such as filtration, centrifugation, or dialysis. This step is critical to prevent interference with downstream analyses.
- 4. Efficacy Validation:
- Microbial Load Reduction: Use standard plate count techniques before and after treatment to quantify the reduction in microbial load (CFU/g).
- Structural Integrity: Use Scanning Electron Microscopy (SEM) to confirm that the treatment caused minimal damage to the starch granules and overall structure of the flour.
- In Vitro Fermentation: Perform simulated colon fermentations and analyze short-chain fatty acid production and microbial composition to confirm that the treatment did not alter the fermentability of the substrate.
Mechanisms of Action: A Visual Guide to Non-Thermal Technologies
The efficacy of non-thermal technologies stems from their distinct mechanisms of action, which target microbial and cellular structures without the widespread molecular damage caused by heat. The following diagram illustrates the primary mechanisms of four key technologies and how they synergize to enhance efficacy.
The Scientist's Toolkit: Essential Research Reagents and Materials
Implementing non-thermal processing in a research setting requires specific reagents and materials. The following table details key items for the experimental protocols discussed in this guide.
Table 3: Essential Research Reagents and Materials for Non-Thermal Processing Experiments
| Item Name | Function/Application | Example/Specification |
|---|---|---|
| Pulse Flours | Model substrate for studying non-thermal effects on starchy, heat-labile materials. | Raw flours from green lentil (Lens culinaris), chickpea (Cicer arietinum), or field pea (Pisum sativum) [70]. |
| Germicidal Bleach | Liquid chemical sterilant for non-thermal microbial load reduction on solid substrates. | Sodium hypochlorite solution, diluted to 1.0-1.5% for treatment [70]. |
| Hydrogen Peroxide | Liquid chemical sterilant; an alternative to bleach for non-thermal treatment. | 30% stock solution, diluted to 2.0% for treatment [70]. |
| Reagent Alcohol | Liquid disinfectant; less effective as a sporicidal agent but accessible. | 90% stock, diluted to 70% ethanol for treatment [70]. |
| High-Pressure Processing Vessel | Core equipment for applying HPP treatments to samples. | Laboratory-scale HPP unit capable of pressures of 100-800 MPa [17]. |
| PEF Treatment Chamber | Core component for applying pulsed electric fields to liquid or semi-solid samples. | A chamber with electrodes connected to a high-voltage pulse generator (e.g., 10-80 kV/cm) [17]. |
| Chromatography System | For quantifying specific bioactive compounds (e.g., aflatoxins, phenolics) post-treatment. | High-Performance Liquid Chromatography (HPLC) system with appropriate detectors (e.g., fluorescence, UV) [70]. |
| Simulated Colon Fermentation Media | For evaluating the impact of non-thermal processing on the fermentability of substrates by gut microbiota. | Complex growth medium designed to mimic the colonic environment for in vitro studies [70]. |
Analyzing Energy Consumption and Environmental Footprint
The food and beverage industry is a significant global energy consumer and contributor to greenhouse gas (GHG) emissions, accounting for approximately one-third of total emissions worldwide [61]. Conventional thermal processing techniques, while effective for microbial safety, are energy-intensive due to their reliance on sustained heating and cooling [61]. In recent years, non-thermal processing technologies have emerged as promising alternatives, offering potential reductions in energy consumption and environmental footprint while better preserving heat-sensitive bioactive compounds [71] [61]. This comparative analysis examines the energy profiles and environmental impacts of thermal and non-thermal food processing technologies, providing researchers and scientists with quantitative data and methodological approaches for objective assessment.
Understanding these parameters is crucial for the development of sustainable food processing systems that align with global sustainability goals, including the United Nations Sustainable Development Goals (SDGs) related to affordable and clean energy, responsible consumption and production, and climate action [72]. The measurement of circularity through Life Cycle Assessment (LCA) and technoeconomic analysis (TEA) provides a scientific basis for evaluating novel technologies and business models in the food industry [61] [72].
Comparative Analysis of Energy Consumption and Carbon Footprint
Quantitative Comparison of Processing Technologies
Table 1: Energy Consumption and Environmental Footprint of Food Processing Technologies
| Processing Technology | Energy Consumption | Carbon Footprint | Primary Energy Source | Key Environmental Advantages |
|---|---|---|---|---|
| Thermal Pasteurization | High (baseline) | Baseline for comparison | Fossil fuels (primarily) | Established, reliable technology |
| High-Pressure Processing (HPP) | Lower than thermal for equivalent shelf life [61] [73] | Comparable to or lower than thermal [61] | Electricity | Water as pressure-transmitting medium, no chemical additives required [73] |
| Pulsed Electric Field (PEF) | Up to 50% lower than thermal methods [43] | Lower due to reduced energy use [43] | Electricity | Minimal water requirements, waste-free processing [71] [43] |
| Cold Plasma (CP) | Highly energy efficient [71] [2] | Not quantified in studies | Electricity | Reduces chemical use, limits waste generation, low water consumption [71] [2] |
| Ultrasonication (US) | Low energy consumption [71] [2] | Not quantified in studies | Electricity | Environmentally friendly, non-toxic, no chemical additives needed [71] [2] |
Table 2: Bioactive Compound Retention Across Processing Technologies
| Processing Technology | Vitamin C Retention | Polyphenol Retention | Antioxidant Capacity | Overall Bioactivity Preservation |
|---|---|---|---|---|
| Thermal Pasteurization | Significant degradation [7] [68] | Moderate to significant degradation | Reduced | Limited preservation of heat-sensitive compounds |
| High-Pressure Processing (HPP) | High retention [71] | High retention [71] [64] | Well preserved [71] [7] | Excellent for most bioactive compounds |
| Pulsed Electric Field (PEF) | High retention (e.g., in orange juice) [7] | Increased (e.g., total polyphenols 10.03% in orange juice) [7] | Enhanced (e.g., 51.10% by ORAC in orange juice) [7] | Superior retention and enhancement |
| Cold Plasma (CP) | Well preserved [71] | Well preserved [71] | Maintained | Good preservation with minimal damage |
| Ultrasonication (US) | High retention [71] [2] | High retention [71] | Maintained or enhanced | Good to excellent preservation |
Analysis of Environmental and Bioactivity Metrics
The comparative data reveals a clear trend: non-thermal technologies generally offer advantages in both energy efficiency and bioactive compound preservation. HPP and PEF demonstrate particularly favorable profiles, with HPP achieving microbial inactivation through uniform hydrostatic pressure (100-900 MPa) without significant heat, thereby preserving covalent bonds in small molecules responsible for bioactive properties [64] [2]. PEF utilizes short bursts of high-voltage electricity (20-80 kV cm⁻¹) to disrupt microbial cell membranes through electroporation, achieving microbial inactivation with minimal temperature increase [64] [43].
The carbon footprint of non-thermal processes is comparable to or lower than thermal pasteurization, with HPP being particularly notable for its waste-free process that eliminates the need for package pre-sterilization and recycles its water-based pressure-transmitting medium [61] [73]. PEF technology stands out for its rapid, continuous processing capability, reducing energy consumption by up to 50% compared to thermal methods while preserving nutrients, flavors, and colors [43].
Experimental Protocols for Energy and Bioactivity Assessment
Life Cycle Assessment (LCA) Methodology
Goal and Scope Definition: Clearly define the assessment objectives, system boundaries (cradle-to-gate or cradle-to-grave), and functional unit (e.g., per liter of processed juice) [61]. For comparative studies, the functional unit must be identical across all technologies compared.
Inventory Analysis: Collect primary data on energy and material inputs/outputs for each processing technology [61]. Key parameters include:
- Energy consumption (kWh per functional unit) from electricity and thermal sources
- Water usage throughout the processing cycle
- Chemical inputs including cleaning agents and preservatives
- Packaging materials and their life cycle impacts
- Emissions to air, water, and soil
- Waste generation and management pathways
Impact Assessment: Evaluate the potential environmental impacts using established methods (e.g., ISO 14040/14044) [61]. Core impact categories should include:
- Global warming potential (carbon footprint in kg CO₂ equivalent)
- Primary energy demand (renewable and non-renewable sources)
- Water consumption (liters of freshwater use)
- Acidification potential (kg SO₂ equivalent)
- Eutrophication potential (kg PO₄ equivalent)
Interpretation: Analyze results to identify environmental hotspots, conduct sensitivity analyses, and draw conclusions about comparative environmental performance [61].
Bioactive Compound Analysis Protocol
Sample Preparation: Process identical food matrices (e.g., orange juice, fruit purees) using thermal and non-thermal technologies with equivalent microbial inactivation efficacy (e.g., 5-log reduction of target pathogens) [7] [64].
Extraction Procedures:
- Polyphenols: Use methanol/water or ethanol/water extraction (typically 70:30 v/v) with sonication at 40°C for 15 minutes [7]
- Vitamins: Employ stabilization with metaphosphoric acid followed by HPLC analysis [7]
- Carotenoids: Extract with hexane:acetone:ethanol solvent system (50:25:25 v/v) under dim light [7]
Analytical Techniques:
- HPLC-DAD/MS for identification and quantification of individual bioactive compounds [7]
- Spectrophotometric assays for total phenolic content (Folin-Ciocalteu method) and antioxidant capacity (DPPH, ORAC, FRAP) [7]
- Bioaccessibility assessment using in vitro gastrointestinal digestion models [7]
Statistical Analysis: Apply multivariate statistical methods (ANOVA, PCA) to identify significant differences (p<0.05) in bioactive compound retention between processing technologies [7].
Visualizing the Comparative Assessment Framework
Technology assessment workflow for comparing processing technologies.
The Researcher's Toolkit: Essential Reagents and Materials
Table 3: Essential Research Reagents and Materials for Comparative Studies
| Reagent/Material | Function/Application | Specific Examples |
|---|---|---|
| Oxygen Radical Absorbance Capacity (ORAC) Assay Kit | Quantifies antioxidant capacity against peroxyl radicals | Commercial ORAC assay kits with fluorescein probe and AAPH radical generator [7] |
| Folin-Ciocalteu Reagent | Measures total phenolic content through phosphomolybdate reduction | Folin-Ciocalteu phenol reagent with gallic acid standard curve [7] |
| DPPH (2,2-Diphenyl-1-picrylhydrazyl) | Assesses free radical scavenging activity | Methanolic DPPH solution with Trolox standard [7] |
| HPLC Standards | Identification and quantification of specific bioactive compounds | Vitamin C, phenolic acids (chlorogenic, ferulic, caffeic), flavonoids (rutin), anthocyanins [7] |
| In vitro Digestion Model Components | Simulates gastrointestinal conditions for bioaccessibility | Enzymes (pepsin, pancreatin, bile extracts), pH modifiers [7] |
| Microbial Culture Media | Validates microbial inactivation efficacy | Plate count agar, selective media for pathogens [64] |
The comprehensive analysis of energy consumption and environmental footprint demonstrates that non-thermal processing technologies offer significant advantages over conventional thermal methods in terms of energy efficiency, carbon footprint reduction, and bioactive compound preservation. While initial investment costs remain higher for technologies like HPP and PEF, their long-term sustainability benefits and superior product quality present compelling cases for adoption, particularly for high-value functional foods and beverages [61] [68] [43].
Future research should focus on standardizing assessment methodologies, exploring synergistic effects of technology combinations, and developing integrated sustainability metrics that encompass environmental, economic, and social dimensions. Such efforts will advance the food industry's transition toward circular economy models and contribute to achieving global sustainability targets while meeting consumer demand for minimally processed, nutrient-rich foods [72].
Evidence-Based Comparison: Measuring Bioactivity Retention and Functional Outcomes
The growing consumer demand for fresh-like, nutritious food products has accelerated the adoption of non-thermal preservation technologies within the fruit processing industry. High-Pressure Processing (HPP) has emerged as a key alternative to conventional Thermal Treatment (TT), promising enhanced retention of bioactive compounds. This case study provides a systematic comparison of the effects of HPP and TT on the stability of polyphenols in strawberry and apple products, framing this analysis within the broader research on how processing technologies impact food bioactivity. Polyphenols, including flavonols, anthocyanins, proanthocyanidins, and phenolic acids, are key bioactive compounds responsible for the antioxidant capacity and associated health benefits of these fruits [74] [75]. Their stability during processing and storage is a critical determinant of the final product's nutritional and functional quality, making them a primary focus for researchers and product developers aiming to maximize health benefits [76].
Comparative Analysis of Processing Impacts on Key Polyphenols
The impact of HPP and TT varies significantly between strawberry and apple products and is highly dependent on the specific polyphenol family. The tables below summarize the documented effects.
Table 1: Impact of Processing on Major Polyphenols in Strawberry Products
| Polyphenol Family | Examples | Thermal Treatment (TT) Effect | High-Pressure Processing (HPP) Effect | Key Factors Influencing Stability |
|---|---|---|---|---|
| Anthocyanins (ATs) | Pelargonidin-3-glucoside, Cyanidin-3-glucoside | Degradation (5-44%) commonly reported; up to 90% loss after storage [77] [78]. | Generally stable post-processing; minor changes. Significant degradation during storage due to residual PPO/POD activity [77] [79]. | Fruit variety, food matrix pH, ascorbic acid content, residual enzyme activity [77]. |
| Ellagic Acid (EA) & Ellagitannins (ETs) | Ellagic acid conjugates | Stable or increased; no change in EA conjugates reported in some studies [76]. | Increase; significant release from ellagitannins, enhancing bioavailability [74] [77]. | Achene content in products, pressure intensity, storage time [74]. |
| Proanthocyanidins (PACs) | Flavan-3-ol oligomers and polymers | Increase; observed in mild and standard thermal treatments [76]. | Increase; improved extractability from the fruit matrix [74]. | Polymerization degree, fruit tissue disruption. |
Table 2: Impact of Processing on Major Polyphenols in Apple Products
| Polyphenol Family | Examples | Thermal Treatment (TT) Effect | High-Pressure Processing (HPP) Effect | Key Factors Influencing Stability |
|---|---|---|---|---|
| Dihydrochalcones (DHCs) | Phloretin glycosides | Increase; standard thermal treatment increases concentrations [76]. | Increase; mild HPP has a similar positive effect [76]. | Variety, part of the fruit (seeds are rich source). |
| Hydroxycinnamic Acids (HCAs) | Chlorogenic acid | Increase [76]. | Increase [76]. | Improved extractability from the matrix post-processing. |
| Proanthocyanidins (PACs) | Flavan-3-ol oligomers and polymers | Increase [76]. | Increase [76]. | Fruit flesh vs. peel concentration, processing conditions. |
| Flavonols (FOLs) | Quercetin derivatives | Variable; standard thermal treatment increased concentrations, while milder treatments decreased them [76]. | Decrease; observed with mild HPP treatments [76]. | Sensitivity to enzymatic oxidation by residual PPO. |
Underlying Mechanisms and Experimental Evidence
The Role of Enzyme Inactivation
A fundamental difference between TT and HPP lies in their efficacy in inactating endogenous enzymes, particularly polyphenol oxidase (PPO) and peroxidase (POD). These enzymes are key drivers of polyphenol degradation and browning in fruit products [74] [80].
TT is highly effective at inactivating PPO and POD, thereby preventing enzymatic oxidation during storage [74]. In contrast, HPP at typical industrial levels (300-600 MPa) often fails to fully inactivate these enzymes. The residual enzymatic activity in HPP-treated products can trigger oxidation reactions during storage, leading to significant degradation of sensitive polyphenols like anthocyanins and flavonols [74] [78]. Research has shown that the resistance of PPO to these technologies varies by fruit type, with one study finding pear PPO to be the most resistant, followed by apple and then strawberry PPO [80].
The following diagram illustrates the divergent pathways through which TT and HPP affect polyphenol stability, highlighting the critical role of enzyme inactivation.
Detailed Experimental Protocols
To ensure the validity and reproducibility of comparative studies, researchers adhere to specific, controlled protocols for both processing technologies.
Table 3: Characteristic Experimental Protocols for HPP and TT
| Protocol Component | High-Pressure Processing (HPP) | Thermal Treatment (TT) |
|---|---|---|
| Typical Equipment | 30 L HPP unit with water as pressure-transmitting medium (e.g., Avure Technologies) [81]. | Water bath or thermal exchanger for precise temperature control [80]. |
| Standard Parameters | Pressure: 400 - 600 MPa [79] [78].Time: 1.5 - 15 minutes [77] [78].Temperature: 20 - 50 °C (ambient or mildly elevated) [77] [78]. | Temperature: 70 - 100 °C [77].Time: 1 - 15 minutes (e.g., 70°C/2min pasteurization; 90°C/5min) [77] [79]. |
| Sample Preparation | Fruit puree/juice vacuum-packed in high-barrier plastic bags (e.g., OPA/PP) [78] [81]. | Fruit puree/juice packed in similar pouches or sealed containers [80]. |
| Key Measurements | Polyphenol content (HPLC), antioxidant activity (ORAC, DPPH, FRAP), color (Hunter L, a, b*), residual PPO/POD activity, viscosity [74] [78] [81]. | Polyphenol content (HPLC), antioxidant activity, color, vitamin C content, microbial load [74] [79]. |
| Storage Studies | Typically at 4-6 °C for up to 12 weeks, with periodic sampling to monitor polyphenol degradation and enzyme activity [78]. | Similar refrigerated storage conditions to assess shelf-life and stability [77]. |
The Scientist's Toolkit: Key Research Reagents and Materials
Table 4: Essential Reagents and Materials for Polyphenol Stability Research
| Item | Function/Application | Example from Search Results |
|---|---|---|
| Chemical Standards | HPLC quantification and identification of specific polyphenols. | Cyanidin-3-glucoside, pelargonidin-3-glucoside (anthocyanins); chlorogenic acid (HCA); phloretin glycosides (DHCs) [79]. |
| Antioxidant Assay Kits | Quantifying total antioxidant capacity of processed samples. | DPPH (2,2-diphenyl-1-picrylhydrazyl), FRAP (Ferric Reducing Antioxidant Power), ORAC (Oxygen Radical Absorbance Capacity) reagents [75] [79]. |
| Enzyme Activity Assays | Measuring residual activity of spoilage enzymes (PPO/POD) post-processing. | Substrates like pyrogallol for PPO activity; specific buffers for pH-controlled enzyme assays [79] [80]. |
| High-Barrier Packaging | Vacuum packaging samples for HPP and storage to prevent oxidative degradation. | OPA/PP (oriented polyamide/polypropylene) plastic bags with low oxygen permeability [78] [81]. |
| Sample Preservation Chemicals | Stabilizing samples for microstructure or chemical analysis. | FAA solution (Formalin-Acetic Acid-Alcohol) for histological analysis [81]; metaphosphoric acid for vitamin C stabilization [79]. |
This case study demonstrates that the choice between HPP and TT involves a critical trade-off. HPP excels at preserving a fresh-like quality and maximizing the initial content and extractability of most polyphenols immediately after processing. However, its inability to consistently fully inactivate endogenous enzymes like PPO and POD can lead to significant degradation of certain sensitive polyphenols, such as anthocyanins in strawberries, during storage. In contrast, while TT may cause more initial degradation of heat-labile compounds, its effective enzyme inactivation ensures greater long-term stability of the polyphenol profile throughout the product's shelf life.
The optimal processing strategy is not universal but depends on the primary fruit (strawberry vs. apple), the specific polyphenol families of interest, the desired shelf life, and the target product's quality attributes. For manufacturers, this underscores the necessity of customizing processing conditions based on the specific fruit and its unique polyphenolic composition to successfully deliver products with enhanced health benefits. For researchers, it highlights the importance of conducting storage stability studies alongside immediate post-processing analysis to fully understand the implications of using these technologies.
Quantifying Retention of Antioxidant Capacity and Vitamin Content
The processing of food is essential for ensuring safety and extending shelf life. However, conventional thermal methods often degrade heat-sensitive nutrients, reducing the nutritional and bioactive value of the final product. In response, non-thermal technologies have emerged as promising alternatives for preserving bioactive compounds. This guide provides a quantitative comparison of thermal and non-thermal processing methods, focusing on their impact on antioxidant capacity and vitamin content. It is designed to support researchers and product development specialists in making evidence-based decisions for preserving bioactivity in food and nutraceutical products.
Quantitative Comparison of Processing Technologies
The following tables consolidate experimental data from scientific studies, providing a direct comparison of how different processing technologies affect key nutritional parameters.
Table 1: Retention of Antioxidant Compounds and Capacity After Processing
| Product | Processing Technology | Conditions | Total Phenolic Content (TPC) | Total Flavonoid Content (TFC) | Total Anthocyanin Content (TAC) | Antioxidant Capacity (DPPH) | Citation |
|---|---|---|---|---|---|---|---|
| Mulberry Vinegar | Thermal Processing (TP) | 85°C, 15 min | Baseline | Baseline | Baseline (Control = Highest) | Baseline | [82] |
| Microwave Heating (MH) | 600W, 40s | < TP | < TP | < TP | < TP | [82] | |
| High Hydrostatic Pressure (HHP500) | 500 MPa, 25°C, 15 min | Highest | Highest | Notable levels | Best Retained | [82] | |
| Orange Juice | Thermal Pasteurization | Not Specified | Lower | N/A | N/A | 38.21% decrease | [83] |
| Pulsed Electric Field (PEF) | Not Specified | Higher than Pasteurized | N/A | N/A | Better preservation than Pasteurization | [83] | |
| High Hydrostatic Pressure (HHP) | Not Specified | Higher than Pasteurized | N/A | N/A | Better preservation than Pasteurization | [83] | |
| Fruit/Vegetable By-products | Ultrasound, Light Stress, Enzyme-assisted | Varies | Increased vs. conventional | N/A | N/A | N/A | [7] |
Table 2: Retention of Vitamin Content After Processing
| Product | Processing Technology | Vitamin C Retention | Carotenoid/Provitamin A Retention | Other Vitamin Notes | Citation |
|---|---|---|---|---|---|
| Orange Juice | Thermal Pasteurization | 82.4% | Decreased by 12.8% | N/A | [83] |
| Pulsed Electric Field (PEF) | 87.5 - 98.2% | N/A | N/A | [83] | |
| High Hydrostatic Pressure (HHP) | 93% | Decreased by only 4.2% | N/A | [83] | |
| Various Vegetables | Boiling | Lowest retention (as low as 0% in some samples) | Varies by vegetable; occasionally higher than raw | Greatest loss of Vitamin K in crown daisy and mallow | [10] |
| Steaming | Moderate retention | Varies by vegetable; occasionally higher than raw | N/A | [10] | |
| Microwaving | Highest retention (up to 91.1%) | Varies by vegetable; occasionally higher than raw | Caused least loss of Vitamin K in spinach and chard | [10] | |
| Cereals & Legumes | Heat Processing (Baking) | N/A | N/A | Significant reductions in B-group vitamins and minerals (e.g., Cu decreased by 82.5% in wheat) | [84] |
Experimental Protocols for Key Studies
To ensure the reproducibility of these findings, the following section details the methodologies employed in the cited comparative studies.
Protocol: Comparison of Sterilization Techniques on Mulberry Vinegar
This study directly compared thermal, microwave, and high-pressure sterilization on a high-acidity fruit product [82].
- Sample Preparation: High-acidity mulberry vinegar was produced via lactic acid fermentation (with Lactobacillus plantarum at 37°C for 44 h) followed by alcoholic fermentation (with Saccharomyces cerevisiae at 22°C) and acetic acid fermentation (with Acetobacter aceti).
- Experimental Treatments:
- Thermal Processing (TP): Samples were heated at 85°C for 15 minutes.
- Microwave Heating (MH): Samples were treated in a microwave at 600 W for 40 seconds.
- High Hydrostatic Pressure (HHP): Samples were subjected to 400, 500, or 600 MPa at ambient temperature (~25°C) for 15 minutes.
- Analysis of Bioactive Compounds:
- Total Phenolic Content (TPC) and Total Flavonoid Content (TFC): Measured using spectrophotometric methods with the Folin-Ciocalteu reagent and aluminum chloride method, respectively.
- Total Anthocyanin Content (TAC): Determined using the pH-differential method.
- Antioxidant Capacity: Assessed via the DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging assay.
- Individual Phenolic Acids: Quantified using High-Performance Liquid Chromatography (HPLC).
Protocol: Analysis of Vitamin Retention in Processed Orange Juice
This research quantified the degradation of heat-sensitive nutrients in orange juice under different preservation methods [83].
- Sample Processing:
- Non-thermal: Orange juice was processed using Pulsed Electric Field (PEF) and High Hydrostatic Pressure (HHP) at non-thermal conditions.
- Thermal: Juice was subjected to conventional thermal pasteurization.
- All samples were subsequently stored under refrigerated conditions (4°C).
- Analytical Methods:
- Vitamin C: Concentration was analyzed, likely via HPLC or a similar chromatographic method.
- Total Carotenoids: Content and profile were determined using liquid chromatography with an ultraviolet-diode array detector (HPLC-UV-DAD).
- Total Phenolic Compounds: Measured using a spectrophotometric method.
- Total Antioxidant Capacity: Evaluated using the Trolox Equivalent Antioxidant Capacity (TEAC) assay.
Protocol: Assessing Cooking Methods on Vegetable Vitamins
This study evaluated the true retention of vitamins in vegetables, accounting for weight changes after cooking [10].
- Cooking Methods: Ten different vegetables were processed using blanching, boiling, microwaving, and steaming under controlled conditions (see Table 1 in [10] for specific times).
- True Retention Calculation: Retention was calculated using the formula:
(Nutrient content per g of cooked food × Weight of cooked food) / (Nutrient content per g of raw food × Weight of raw food) × 100. This accounts for concentration or dilution effects due to water loss or absorption. - Vitamin Analysis:
- Vitamin C: Analyzed by HPLC with a UV detector after extraction with metaphosphoric acid.
- Fat-Soluble Vitamins (E, K, β-carotene): Quantified using HPLC with fluorescence or UV detection after saponification extraction or solvent extraction.
Workflow for Comparative Analysis
The diagram below outlines a generalized experimental workflow for conducting a comparative analysis of food processing technologies, synthesizing the protocols from the cited studies.
The Scientist's Toolkit: Essential Research Reagents and Materials
This table lists key reagents, materials, and equipment essential for conducting research on nutrient retention in food processing, as derived from the experimental protocols.
Table 3: Research Reagent Solutions and Essential Materials
| Item | Function/Application | Example from Context |
|---|---|---|
| Folin-Ciocalteu Reagent | Spectrophotometric quantification of total phenolic content (TPC). | Used in analysis of mulberry vinegar [82]. |
| DPPH (2,2-diphenyl-1-picrylhydrazyl) | A stable free radical used to assess antioxidant capacity via radical scavenging assays. | Used to determine antioxidant activity in mulberry vinegar and fruit studies [82] [75]. |
| HPLC-Grade Standards | Reference compounds for identification and quantification via chromatography. | Phenolic acids, vitamins (C, E, K), carotenoids, and amino acids for accurate HPLC analysis [82] [10]. |
| Metaphosphoric Acid | A stabilizing agent used in the extraction of vitamin C to prevent its degradation during analysis. | Used in vegetable vitamin analysis before HPLC injection [10]. |
| Pectinase | An enzyme used to break down pectin, clarifying fruit pulps and juices before fermentation or analysis. | Used in the pretreatment of mulberry fruit for vinegar production [82]. |
| Specific Microorganism Strains | Used in fermentation studies to produce model food systems (e.g., vinegar, fermented beverages). | Lactobacillus plantarum, Saccharomyces cerevisiae, Acetobacter aceti [82]. |
| High-Pressure Processing (HPP) Unit | Equipment for applying high hydrostatic pressure for non-thermal processing/preservation. | Used to process samples at 400-600 MPa [82] [2]. |
| Pulsed Electric Field (PEF) System | Equipment for applying short, high-voltage pulses for non-thermal microbial inactivation. | Used for juice processing [83]. |
Impact on Bioaccessibility and Bioavailability for Human Nutrition
The processing of food is a critical determinant of its nutritional value, influencing the release, absorption, and ultimate utilization of bioactive compounds by the human body. While thermal processing has been the cornerstone of food preservation for centuries, its application can lead to the degradation of heat-sensitive nutrients, potentially diminishing their health-promoting properties [16] [17]. In response to growing consumer demand for fresh-like, nutritious foods, non-thermal processing technologies have emerged as promising alternatives [2] [60]. This guide provides an objective comparison of thermal and non-thermal processing methods, focusing on their distinct impacts on the bioaccessibility and bioavailability of dietary bioactives. It is structured to support researchers, scientists, and product developers in making evidence-based decisions for nutritional optimization.
Bioaccessibility refers to the proportion of a compound that is released from the food matrix and becomes available for intestinal absorption, while Bioavailability describes the fraction that is absorbed, enters systemic circulation, and is utilized for physiological functions [85]. The journey of a bioactive compound from ingestion to utilization involves several stages, and processing methods can profoundly influence each step.
Comparative Analysis of Processing Technologies
Non-Thermal Processing Technologies
Non-thermal technologies inactivate microorganisms and enzymes with minimal heat application, thereby preserving heat-sensitive nutrients. Their mechanisms range from physical disruption of cell structures to chemical oxidation.
- High-Pressure Processing (HPP): This method subjects food, within a sealed chamber, to intense hydrostatic pressure (typically 100-600 MPa). HPP operates on the isostatic principle (pressure is instantaneously and uniformly distributed) and Le Chatelier's principle (pressure favors reactions that result in a volume decrease) [2] [60]. It inactivates microbes by disrupting non-covalent bonds and collapsing cellular structures, while leaving small molecules like vitamins and pigments largely intact [2].
- Pulsed Electric Field (PEF): PEF applies short, high-voltage pulses (e.g., 20-80 kV/cm) to a food product placed between two electrodes [17]. Its primary mechanism is electroporation, where the electric field induces pore formation in the membranes of microbial and plant cells [17]. This can lead to microbial inactivation and, importantly, enhance the release of intracellular bioactives from the food matrix, thereby increasing their bioaccessibility [26].
- Cold Plasma (CP): Cold plasma is a partially ionized gas generated at low temperatures, containing a mix of reactive species (ions, electrons, radicals). These reactive molecules inactivate microorganisms through oxidative damage to cell membranes and genetic material. CP is also effective in degrading pesticide residues and mycotoxins, improving food safety [2].
- Ultrasound (US): Ultrasonication uses high-frequency sound waves to create cavitation bubbles in a liquid medium. The implosion of these bubbles generates intense local shear forces, microjets, and shock waves. This physical force disrupts cell walls, facilitating the release of bioactives, and can also reduce microbial load [60].
Thermal Processing Technologies
Thermal processing relies on heat to achieve microbial safety and shelf-life extension. The effects on bioactives are highly dependent on temperature, time, and the specific food matrix [16].
- Mechanisms and Variable Outcomes: Heat can induce both desirable and undesirable changes. It denatures proteins, which may improve digestibility by unfolding their structure and exposing cleavage sites for enzymes [86]. It can also inactivate antinutritional factors and, in some cases, increase the extractability of certain phenolic compounds from plant tissues [16]. However, excessive heat can lead to the degradation of thermolabile vitamins (e.g., Vitamin C), phenolic compounds, and carotenoids. It can also promote the formation of protein aggregates and oxidation products that are resistant to digestive enzymes, potentially reducing protein digestibility [16] [86].
- Conventional Heat Treatments: Methods such as boiling, steaming, baking, and frying are widely used. For instance, a study on sardines and sprats found that all these thermal methods enhanced protein digestibility compared to raw fish, with frying yielding the highest digestibility (up to 92.4%) but raising potential health concerns due to associated lipid oxidation [86].
- Microwave Processing: A specific form of thermal processing, microwave heating, has been shown to be highly effective at reducing antinutrients and pesticide residues. For example, in potato powder, microwave treatment reduced alkaloids by 76% and tannins by 84%, while also degrading pesticides like imidacloprid by 87% [87].
Quantitative Comparison of Processing Effects
The following tables synthesize experimental data from recent studies to provide a direct comparison of the effects of thermal and non-thermal processing on various bioactive compounds.
Table 1: Impact of Thermal vs. Non-Thermal Processing on Fruit Juice Bioactives (Fruit Juice Blend: Kiwi, Mango, Orange, Blueberry) [26]
| Processing Method | Specific Conditions | Total Phenolic Content (TPC) | Total Anthocyanin Content (TAC) | Antioxidant Capacity | Key Findings |
|---|---|---|---|---|---|
| High-Pressure Processing (HPP) | 600 MPa / 3 min | Highest value among tested conditions | High | Highest value | Optimal condition for retaining bioactive substance and antioxidant capacity. |
| Pulsed Electric Field (PEF) | 120 kJ/L - 24 kV/cm | Highest value among tested conditions | High | Highest value | Optimal condition for retaining bioactive substance and antioxidant capacity. |
| Thermal Treatment (TT) | 80 °C / 30 min | Lower than HPP & PEF | Lower than HPP & PEF | Lower than HPP & PEF | Resulted in lower retention of bioactive compounds compared to optimal non-thermal methods. |
| Post-Digestion Analysis | PEF treated samples | Highest | Highest | N/D | PEF treatment resulted in the highest bioaccessibility for TPC, TFC, and TAC after in vitro digestion. |
Table 2: Impact of Drying Methods on Flavonoid Retention in Loquat Flowers [88]
| Processing Method | Specific Conditions | Impact on Key Flavonoids | Antioxidant Activity (μg TE/g) | Key Findings |
|---|---|---|---|---|
| Freeze-Drying (Non-Thermal) | -50°C under vacuum | Cyanidin: 6.62-fold increase vs HDDelphinidin: 49.85-fold increase vs HD | 608.83 (Freeze-Dried Powder) | Optimal for thermolabile compounds. Significantly preserved a wide range of flavonoids, leading to the highest antioxidant activity. |
| Heat-Drying (Thermal) | 60°C for 6 hours | 6-Hydroxyluteolin: 27.36-fold increaseMany other flavonoids degraded | Lower than Freeze-Dried | Selective enhancement of a few heat-stable flavonoids, but overall degradation of many thermolabile compounds. |
Table 3: Effect of Thermal Processing on Protein Digestibility in Fish [86]
| Processing Method | Conditions | Total Protein Digestibility (Sardines) | Total Protein Digestibility (Sprats) | Key Findings |
|---|---|---|---|---|
| Raw (Control) | N/A | Baseline | Baseline | Baseline digestibility. |
| Boiling | 100°C, 10 min | Increased vs. Raw | Increased vs. Raw | Improved digestibility through protein denaturation. |
| Steaming | 100°C, 20 min | Increased vs. Raw | Increased vs. Raw | Improved digestibility; favorable balance with lower health risks. |
| Baking | 180°C, 20 min | Increased vs. Raw | Increased vs. Raw | Comparable improvement to steaming and boiling. |
| Frying | 180°C, 5 min in oil | 92.4 ± 4.3% (Highest) | 89.5 ± 4.4% (Highest) | Yielded the highest digestibility and DIAAS scores, though associated with lipid oxidation. |
Detailed Experimental Protocols
To ensure reproducibility and a deep understanding of the data presented, this section outlines key methodologies from the cited research.
Protocol 1: Investigating Processing Effects on Fruit Juice Blend
This protocol is adapted from the study on a fruit juice blend comprising kiwi, mango, orange, and blueberry [26].
- 1. Sample Processing:
- HPP Treatment: Apply treatments at various pressure and time combinations, including 600 MPa for 3 minutes, 500 MPa for 5 minutes, and 500 MPa for 10 minutes.
- PEF Treatment: Apply treatments at various energy input and field strength combinations, including 120 kJ/L at 24 kV/cm, 100 kJ/L at 20 kV/cm, and 100 kJ/L at 15 kV/cm.
- Thermal Treatment: Treat the juice blend at 80°C for 30 minutes.
- 2. Bioactive Compound Extraction: Homogenize processed juice samples and use aqueous/organic solvent mixtures (e.g., methanol/water) to extract phenolic compounds, anthocyanins, and other antioxidants.
- 3. In Vitro Digestion Simulation: Subject the processed juices to a simulated gastrointestinal digestion model.
- Oral Phase: Mix with simulated saliva fluid (containing α-amylase) for a short period.
- Gastric Phase: Adjust to pH 3.0 with HCl and add pepsin, incubate for 1-2 hours at 37°C.
- Intestinal Phase: Adjust to pH 7.0 with NaOH, add pancreatin and bile salts, incubate for 2+ hours at 37°C.
- 4. Analytical Measurements:
- Total Phenolic Content (TPC): Use the Folin-Ciocalteu assay, expressing results as mg Gallic Acid Equivalents (GAE) per 100g or mL.
- Total Anthocyanin Content (TAC): Use the pH-differential method with spectrophotometric measurement.
- Antioxidant Capacity: Assess using assays such as FRAP (Ferric Reducing Antioxidant Power) or ABTS/DPPH radical scavenging, expressing results as Trolox Equivalents (TE).
Protocol 2: Metabolomic Analysis of Processed Loquat Flowers
This protocol details the comprehensive approach used to compare drying methods on loquat flowers [88].
- 1. Sample Preparation and Drying:
- Heat-Drying (HD): Dry freshly harvested loquat flowers in a controlled temperature oven at 60°C for 6 hours until constant weight.
- Freeze-Drying (FD): Flash-freeze flowers at -20°C or -80°C, then lyophilize in a vacuum freeze-dryer (e.g., Scientz-100F) at -50°C for 48 hours.
- 2. Powdered Extract Preparation: Prepare hot-water extracts from both HD and FD flowers using a 1:20 (w/v) biomass-to-solvent ratio at 90°C for 30 minutes. Centrifuge, collect the supernatant, and freeze-dry to obtain a stable powder.
- 3. Flavonoid Profiling via UPLC-MS/MS:
- Metabolite Extraction: Grind dried samples to a fine powder. Weigh 30 mg and add 1.5 mL of pre-cooled 70% methanol-water solution containing internal standards (e.g., 2-chlorophenylalanine). Vortex vigorously, centrifuge, and filter the supernatant through a 0.22 μm membrane.
- Chromatographic Separation: Use an UPLC system (e.g., ExionLC AD) with a C18 column (e.g., Agilent SB-C18, 1.8 μm, 2.1 mm × 100 mm). Employ a mobile phase gradient of water with 0.1% formic acid (A) and acetonitrile with 0.1% formic acid (B).
- Mass Spectrometry Analysis: Interface the UPLC with a triple quadrupole mass spectrometer (e.g., QTRAP 6500+) operating in multiple reaction monitoring (MRM) mode for high sensitivity and specificity.
- 4. Data Analysis: Process the raw MS data using analytical software (e.g., MetWare Database). Perform multivariate statistical analyses, including Principal Component Analysis (PCA) and Orthogonal Projections to Latent Structures-Discriminant Analysis (OPLS-DA), to identify differentially accumulated flavonoids.
Pathways and Mechanisms of Bioactive Modification
The following diagrams illustrate the logical workflow for evaluating processing effects and the subsequent biological fate of bioactives, highlighting critical decision points and mechanisms.
Figure 1. Logical workflow for evaluating the impact of food processing on bioactive compounds. The diagram contrasts the fundamental mechanisms of thermal and non-thermal technologies, leading to distinct alterations in the food matrix and, consequently, different outcomes in the bioaccessibility and bioavailability of bioactive compounds.
Figure 2. The biological fate of bioactive compounds from ingestion to utilization. The diagram distinguishes between bioaccessibility (the release of compounds in the gut) and bioavailability (the absorption and entry into systemic circulation), highlighting key physiological barriers and processes like hepatic metabolism that ultimately determine the efficacy of a nutrient.
The Scientist's Toolkit: Research Reagent Solutions
This section details essential materials and reagents used in the featured experiments, providing a quick reference for researchers aiming to replicate or design similar studies.
Table 4: Essential Reagents and Equipment for Bioaccessibility Research
| Item Name | Function/Application | Example from Research Context |
|---|---|---|
| Folin-Ciocalteu Reagent | Chemical assay for quantifying total phenolic content (TPC) based on a reduction-oxidation reaction. | Used to measure the total phenolic content in fruit juice blends after HPP, PEF, and thermal processing [26]. |
| UPLC-MS/MS System | Ultra-Performance Liquid Chromatography coupled with Tandem Mass Spectrometry for high-resolution separation, identification, and quantification of metabolites. | Used for comprehensive flavonoid profiling in loquat flowers to compare heat-drying vs. freeze-drying [88]. |
| In Vitro Digestion Model | Simulated physiological fluids (saliva, gastric, intestinal) to predict the bioaccessibility of compounds without human trials. | Used to determine the bioaccessibility of phenolic compounds and anthocyanins in processed fruit juices [26] [85]. |
| Standard Compounds (GAE, TE) | Gallic Acid Equivalents (GAE) and Trolox Equivalents (TE) are used as calibration standards for phenolic and antioxidant assays, respectively. | Essential for generating quantitative data in the Folin-Ciocalteu and FRAP/ABTS/DPPH assays [26] [16]. |
| Simulated Gastrointestinal Enzymes | Purified enzymes (α-amylase, pepsin, pancreatin) and bile salts used to mimic human digestion in in vitro models. | Critical components of the simulated digestion protocol to hydrolyze food matrices and assess compound release [26] [85]. |
| Freeze-Dryer (Lyophilizer) | Equipment that removes water from a frozen sample under vacuum, preserving the structure and stability of heat-sensitive compounds. | Used to prepare stable, powdered samples of loquat flowers and their extracts for metabolomic analysis [88]. |
Shelf-life is a critical parameter defining the period during which a food product remains safe, complies with nutritional declarations, and retains its desired sensory, chemical, and physical characteristics under recommended storage conditions [89]. For most food products, shelf-life is determined not by safety concerns but by changes in sensory characteristics that render them unacceptable to consumers [89]. This comparative assessment examines how thermal and non-thermal processing technologies influence sensory quality and shelf-life, with particular emphasis on implications for bioactive compound preservation.
The growing consumer demand for fresh, safe, and high-quality foods with clean labels has driven research into processing technologies that extend shelf-life while minimizing damage to nutritional and sensory properties [90]. While thermal processing remains the most conventional and cost-effective method for ensuring microbial safety, it often damages heat-sensitive bioactive compounds and can produce undesirable sensory changes [7] [90]. Non-thermal technologies have emerged as alternatives that can effectively preserve sensory attributes while maintaining microbial safety, thus offering significant potential for enhancing product shelf-life without compromising quality [90] [91].
Fundamental Principles of Sensory Shelf-Life Determination
Defining Sensory Shelf-Life
Sensory shelf-life (SSL) refers to the period during which a food product maintains its organoleptic characteristics within acceptable ranges for consumers [92]. This encompasses taste, aroma, texture, appearance, and overall acceptability. The determination of SSL is inherently consumer-centric, as technical measurements of quality alone cannot predict rejection if consumers find altered sensory characteristics undesirable [92]. Research has consistently demonstrated a high correlation between consumers' perception of quality and their acceptance of food products, establishing consumer perception as the ultimate criterion for SSL determination [92].
Methodological Approaches for SSL Estimation
Several methodological approaches exist for estimating sensory shelf-life, each with distinct applications and limitations:
- Descriptive Tests: These utilize trained panels to measure specific quality changes in products over time, providing detailed sensory profiles but not direct consumer acceptability data [93].
- Difference Testing: Employed when the shelf-life criterion is the first detectable change in a specific attribute or overall difference [93].
- Hedonic Testing: Conducted to evaluate the impact of quality changes on consumer acceptability and preference, typically using acceptability scales [93].
- Survival Analysis: A statistically robust method that models the probability of consumer rejection over time, directly incorporating consumer perception into shelf-life estimation [89] [94].
Current scientific consensus emphasizes the superiority of consumer-based methodologies over those relying solely on trained panels, as failure criteria based on instrumental measurements or expert judgment may not accurately reflect consumer acceptance thresholds [89].
Table 1: Methodologies for Sensory Shelf-Life Estimation
| Method | Key Features | Applications | Limitations |
|---|---|---|---|
| Descriptive Analysis | Trained panelists quantify specific sensory attributes | Tracking intensity changes in key quality parameters over time | Does not directly measure consumer acceptance |
| Difference Testing | Determines point when products become perceptibly different | Identifying first significant change in sensory profile | Does not indicate whether difference affects acceptability |
| Hedonic Testing | Measures consumer liking using acceptability scales | Establishing correlation between storage time and preference | Requires large consumer panels; multiple time points |
| Survival Analysis | Models consumer rejection probability over time | Determining shelf-life based on consumer rejection criteria | Complex experimental design and statistical analysis |
Impact of Processing Technologies on Sensory Quality and Shelf-Life
Thermal Processing Technologies
Conventional thermal processing techniques, including pasteurization, scalding, and drying, effectively ensure microbial safety and enzyme deactivation but frequently cause detrimental effects on sensory and nutritional quality [7]. The heat-sensitive nature of many bioactive compounds and flavor precursors makes them particularly vulnerable to thermal degradation.
In dairy applications, high-temperature long-time (HTLT) treatments of donkey milk whey proteins resulted in significant deterioration of functional and sensory properties. The study reported substantial losses in key bioactive components: lysozyme activity preservation was only 23%, while lactoferrin and IgG suffered losses of 70% and 77%, respectively [8]. These damages to protein structure and functionality inevitably affect sensory characteristics such as mouthfeel, flavor, and appearance, ultimately limiting the shelf-life gains achieved through microbial reduction.
Thermal treatments can also induce the formation of undesirable chemical compounds that negatively impact sensory quality. For instance, conventional thermal processing has been associated with the formation of heterocyclic aromatic amines in meat products and hydroxymethyl-L-furfural in carbohydrate-rich systems, contributing to off-flavors and potential health concerns [90].
Non-Thermal Processing Technologies
Non-thermal technologies process foods at or near ambient temperature, thereby preserving heat-sensitive compounds while effectively reducing microbial loads [90]. These technologies represent a paradigm shift in food processing, aligning with consumer demands for fresh, minimally processed foods with extended shelf-lives.
High-Pressure Processing (HPP): Applied at 400-600 MPa, HPP effectively inactivates microorganisms while preserving nutritional and sensory qualities. In potato varieties, HPP at 600 MPa achieved significant enzyme inactivation while maintaining phenolic content and antioxidant activity [7]. The application of HPP to Burrata cheese extended its sensory shelf-life by mitigating undesirable changes in consistency [89].
Pulsed Electric Fields (PEF): This technology applies short bursts of high voltage to disrupt microbial membranes without significant heating. In carrot-based products, PEF treatment (3.5 kV·cm⁻¹) significantly increased phenolic and carotenoid bio-accessibility, enhancing both nutritional and sensory properties [7]. The technology has proven particularly effective for liquid foods such as juices and beverages.
Ultrasound (US): Utilizing sound waves above 20 kHz, ultrasonication generates cavitation that disrupts microbial cells while potentially improving functional properties. In orange juice, thermo-sonication combined with nisin not only achieved effective microbial inactivation but also increased total polyphenols (10.03%), total carotenoids (20.10%), and antioxidant capacity while largely retaining ascorbic acid and flavonoids [7].
Ultraviolet (UV) Light: This non-thermal treatment acts as both a germicidal agent and an elicitor of beneficial phytochemical accumulation. UV treatment of whole fruits has been shown to induce the accumulation of health-promoting compounds including ascorbic acid, carotenoids, and phenolic compounds, potentially enhancing both sensory and nutritional profiles [7].
Cold Plasma (CP): This emerging technology utilizes ionized gas to surface-pasteurize foods without heat, effectively inactivating pathogens while preserving sensory attributes. CP has shown particular promise for improving the cooking and textural properties of food grains while maintaining their nutritional integrity [90].
Table 2: Comparative Impact of Thermal and Non-Thermal Technologies on Product Quality
| Technology | Sensory Quality Preservation | Bioactive Compound Retention | Typical Shelf-Life Extension |
|---|---|---|---|
| Thermal Processing | Moderate to poor; often causes cooked flavors, texture degradation | Low to moderate; significant loss of heat-labile compounds | Variable; often limited by sensory deterioration |
| High-Pressure Processing | Excellent; minimal effect on fresh characteristics | High; effectively preserves most bioactive compounds | Significant; 2-3 fold extension for many products |
| Pulsed Electric Fields | Very good; minimal sensory changes | High; may enhance bioaccessibility of some compounds | Moderate to significant; particularly effective for liquids |
| Ultrasound | Good; can improve some functional properties | High; may intensify extraction of bioactive compounds | Moderate; often used in combination with other methods |
| UV Light | Excellent; no thermal damage | High to very high; may stimulate biosynthesis of phytochemicals | Moderate; particularly effective for surface treatment |
| Cold Plasma | Excellent; preserves raw characteristics | High; minimal effect on nutritional components | Moderate; mainly for surface decontamination |
Comparative Experimental Data: Thermal vs. Non-Thermal Processing
Dairy Processing Applications
A comparative study on donkey milk whey proteins revealed striking differences between thermal and non-thermal treatments. While thermal processing caused significant deterioration in protein structure and functionality, non-thermal treatments demonstrated superior preservation of bioactive components [8]. Specifically:
- Ultrasonication reduced protein particle size and turbidity, improving physicochemical properties without damaging protein native structure.
- UV treatment oxidized internal disulfide bonds within proteins, increasing free sulfhydryl groups and consequently enhancing foaming and emulsification stability—key parameters influencing sensory characteristics in dairy products [8].
These structural advantages translated to improved functional performance, suggesting better retention of sensory qualities throughout shelf-life.
Fruit and Vegetable Processing
In fruit-based products, non-thermal technologies have demonstrated remarkable effectiveness in preserving sensory and nutritional quality. When applied to orange juice, thermo-sonication with nisin achieved multiple benefits: desirable bactericidal effects, improved physicochemical properties, enhanced sensory quality, and increased bioactive compound content [7]. The combined treatment increased total polyphenols by 10.03%, total carotenoids by 20.10%, and antioxidant capacity by 51.10% (ORAC method) and 10.58% (DPPH method), while largely retaining ascorbic acid and total flavonoids [7].
Similarly, studies on strawberry and apple products indicated that the effect of processing on phenolic content depended on multiple factors including fruit type, polyphenol family, and specific processing conditions [7]. While both thermal and high-pressure processing affected phenolic stability, HPP generally showed superior retention of sensory and nutritional quality parameters.
Meat and Fish Products
Research on fish products confirms that non-thermal technologies can effectively extend shelf-life while maintaining sensory attributes superior to thermal processing [95]. In minced meat, predictive modeling using survival analysis and Arrhenius equations demonstrated that appropriate non-thermal treatments could establish precise shelf-life predictions based on consumer rejection criteria [93]. With 50% consumer rejection as the endpoint, researchers successfully predicted shelf-lives at different storage temperatures, providing a scientifically valid approach to shelf-life determination that directly incorporates consumer perception [93].
Experimental Design for Sensory Shelf-Life Studies
Key Methodological Considerations
Well-designed sensory shelf-life studies require careful planning and execution. The essential steps include [89]:
- Determining Study Objectives: Clearly define whether the study aims to establish initial shelf-life, compare formulations, or evaluate processing technologies.
- Sample Selection and Storage: Ensure samples are representative and storage conditions reflect realistic distribution and consumer handling.
- Evaluation Schedule: Establish appropriate testing intervals based on expected deterioration rate, with more frequent sampling during periods of rapid change.
- Sensory Methodology Selection: Choose appropriate sensory tests (discriminative, descriptive, or affective) based on study objectives.
- Data Analysis and Interpretation: Employ statistical methods appropriate for the experimental design and data type.
Survival Analysis Methodology
Survival analysis has emerged as a particularly robust method for sensory shelf-life estimation as it directly incorporates consumer rejection data. The implementation typically involves [89] [94]:
- Consumer Recruitment: Typically 50-100 consumers representative of the target market.
- Sample Presentation: Samples with varying storage times are presented to consumers in balanced order.
- Acceptability Judgment: Consumers indicate whether they would accept or reject each sample based on sensory properties.
- Data Analysis: Survival statistics model the probability of consumer rejection over time, typically identifying the time at which a predetermined proportion of consumers (often 25% or 50%) would reject the product.
A study on packaged croissants successfully employed survival analysis to determine that 50% of consumers rejected products when they reached specific physicochemical parameters: humidity below 18%, water activity below 0.81, firmness exceeding 1064 N, pH above 4.4, and acidity below 4.5 [94]. This approach established correlations between analytical measurements and consumer acceptance, enabling more accurate shelf-life predictions.
Diagram 1: Sensory Shelf-Life Determination Using Survival Analysis
The Researcher's Toolkit: Essential Methodologies and Reagents
Table 3: Essential Research Tools for Sensory Shelf-Life Studies
| Category | Specific Tools/Methods | Application in SSL Research |
|---|---|---|
| Sensory Evaluation Methods | Quantitative Descriptive Analysis (QDA) | Characterizing specific sensory changes during storage |
| Hedonic Scaling | Measuring consumer liking and acceptability | |
| Survival Analysis | Modeling consumer rejection over time | |
| Physicochemical Analytical Methods | Texture Analysis | Quantifying changes in firmness, hardness, etc. |
| Water Activity Measurement | Monitoring moisture migration and microbial stability | |
| pH and Titratable Acidity | Tracking chemical changes during storage | |
| Chromatography (GC, HPLC) | Analyzing specific compound degradation/formation | |
| Microbiological Assessment | Total Mesophilic Count | Evaluating overall microbial load |
| Specific Pathogen Testing (Salmonella, Listeria, E. coli) | Ensuring safety compliance | |
| Yeast and Mold Counts | Monitoring spoilage microorganisms | |
| Statistical Methods | Analysis of Variance (ANOVA) | Determining significant differences between treatments |
| Survival Statistics | Estimating shelf-life based on consumer rejection | |
| Regression Analysis | Correlating instrumental and sensory data | |
| Accelerated Storage Methods | Arrhenius Modeling | Predicting shelf-life at different temperatures |
| Elevated Temperature Studies | Rapid shelf-life estimation |
The comparative assessment of sensory quality and shelf-life reveals a complex interplay between processing technologies, product stability, and consumer acceptance. While thermal processing remains effective for microbial safety, its detrimental effects on sensory characteristics and bioactive compounds often limit the sensory shelf-life of processed products. Non-thermal technologies present compelling alternatives that can effectively extend shelf-life while preserving, and in some cases enhancing, sensory and nutritional quality.
The determination of sensory shelf-life must ultimately incorporate consumer perception, as analytical measurements alone cannot predict rejection thresholds. Methodologies such as survival analysis provide robust, consumer-centered approaches to shelf-life estimation that align with modern market demands. As food processing continues to evolve, the integration of appropriate non-thermal technologies with scientifically valid shelf-life determination methods will be essential for meeting consumer expectations for safe, high-quality products with extended shelf-lives.
Future research should focus on optimizing combination processing approaches, elucidating the fundamental mechanisms underlying sensory deterioration, and developing more accurate predictive models that incorporate both consumer perception and analytical measurements. Such advances will further enhance our ability to deliver products that maintain superior sensory quality throughout their shelf-life while ensuring safety and nutritional value.
Conclusion
The evidence strongly indicates that non-thermal processing technologies offer a superior pathway for preserving the bioactivity of food compounds critical for health and drug development. While thermal methods ensure safety, they often do so at the cost of degrading heat-sensitive nutraceuticals. Non-thermal techniques like HPP and PEF can effectively inactivate pathogens while better retaining antioxidants, polyphenols, and vitamins, thereby enhancing the functional quality of food matrices. Future research must focus on standardizing protocols, conducting long-term clinical studies to validate health benefits, and developing hybrid processing models. For biomedical research, this promises more reliable plant-based material for drug discovery and the creation of efficacious functional foods, ultimately bridging the gap between food science and clinical health outcomes.
References
- 1. Nutrient Stability and Degradation in Food Processing [https://www.labmanager.com/nutrient-stability-and-degradation-in-food-processing-34284]
- 2. Advances in non-thermal food processing - RSC Publishing [https://pubs.rsc.org/en/content/articlehtml/2025/fb/d5fb00136f]
- 3. Effects of high‐temperature short‐time processing on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9834863/]
- 4. Exploring Formation and Control of Hazards in Thermal ... [https://www.mdpi.com/2304-8158/14/13/2168]
- 5. Non-thermal Technologies for Food Processing - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8217760/]
- 6. Stability and degradation mechanism of (−)-epicatechin in ... [https://www.sciencedirect.com/science/article/pii/S0308814624036884]
- 7. Thermal and Non-Thermal Treatments to Preserve and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9656200/]
- 8. Effect of thermal and nonthermal processing methods on ... [https://www.sciencedirect.com/science/article/pii/S0022030224011020]
- 9. Maillard reaction in different food products: Effect on ... [https://www.sciencedirect.com/science/article/abs/pii/S0956713523003110]
- 10. Effect of different cooking methods on the content ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6049644/]
- 11. Nutritional Quality and Oxidative Stability during Thermal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9030854/]
- 12. Effects of different heat treatments on Maillard reaction ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10063903/]
- 13. Recent progress in the thermal treatment of oilseeds and ... [https://www.sciencedirect.com/science/article/pii/S2667325821001771]
- 14. Thermal and non-thermal processing affect Maillard ... [https://www.sciencedirect.com/science/article/abs/pii/S2212429221001693]
- 15. Alternative Processing Options for Improving the Proteins ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10572503/]
- 16. The current state of knowledge about thermal processing ... [https://www.sciencedirect.com/science/article/pii/S0308814624021769]
- 17. Revisiting Non-Thermal Food Processing and Preservation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8234293/]
- 18. A comprehensive review on impact of non-thermal ... [https://www.sciencedirect.com/science/article/abs/pii/S0963996921005469]
- 19. Frontiers | Sublethal Injury and Viable but Non -culturable (VBNC) State... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2018.02773/full]
- 20. The Effect of Non-Thermal Processing on the Fate ... [https://www.mdpi.com/2304-8158/14/13/2374]
- 21. Novel non technologies | PPT thermal preservation [https://www.slideshare.net/slideshow/novel-non-thermal-preservation-technologies/125507357]
- 22. A Review of Non-thermal Interventions in Food Processing ... [https://www.sciencedirect.com/science/article/pii/S0362028X25000602]
- 23. comparative metabolomic analysis of dried loquat ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12354605/]
- 24. Impact of thermal processing on polyphenols, carotenoids ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11605278/]
- 25. Bioavailability and mechanisms of dietary polyphenols ... [https://www.sciencedirect.com/science/article/pii/S2665927124000418]
- 26. Effects of non-thermal food processing techniques on the ... [https://www.sciencedirect.com/science/article/pii/S2590157525008478]
- 27. The effect of cold plasma treatment on the phenolic and ... [https://www.nature.com/articles/s41598-025-10281-x]
- 28. A Review of the Current Knowledge of Thermal Stability ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8468304/]
- 29. Thermal and non-thermal processing effect on açai juice ... [https://www.sciencedirect.com/science/article/pii/S0963996920305317]
- 30. Household non-thermal processing for the dissolution of ... [https://www.sciencedirect.com/science/article/pii/S2590157525007886]
- 31. High pressure processing of fruit beverages: A recent trend [https://www.sciencedirect.com/science/article/abs/pii/S2949824424000077]
- 32. Effects of High-Pressure Processing (HPP) on Antioxidant ... [https://www.mdpi.com/2076-3417/15/19/10699]
- 33. Benefits of HPP in vegetable foods: preservation of bioactivity [https://www.hiperbaric.com/en/benefits-of-hpp-in-vegetable-foods-preservation-of-bioactivity/]
- 34. Innovative Non-thermal Processing Technologies for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12631679/]
- 35. The Effect of High Pressure Processing on Textural ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9023011/]
- 36. Comparison of high pressure and thermal pasteurization ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10082863/]
- 37. High pressure processing (HPP) vs traditional shelf-stable ... [https://www.hiperbaric.com/en/high-pressure-processing-hpp-vs-traditional-shelf-stable-methods/]
- 38. Effect of traditional thermal and high-pressure processing ... [https://www.sciencedirect.com/science/article/abs/pii/S0023643819303536]
- 39. Pulsed Electric Fields for Food Processing Technology | IntechOpen [https://www.intechopen.com/chapters/38363]
- 40. The application of PEF technology in food processing and human nutrition - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7847884/]
- 41. Comprehensive review on pulsed electric field in food preservation: gaps in current studies for potential future research - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10318501/]
- 42. Evaluation of Pulsed Electric Field and Conventional ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9031134/]
- 43. PEF vs. Conventional Thermal Processing: A Comparative ... [https://www.opticept.se/pef-vs-conventional-thermal-processing-a-comparative-insight/]
- 44. High Pressure Processing (HPP) and Pulsed Electric ... [https://www.hiperbaric.com/en/high-pressure-processing-hpp-and-pulsed-electric-fields-pef-a-comparative-overview-in-juice-processing/]
- 45. Comprehensive review on pulsed electric field in food ... [https://www.sciencedirect.com/science/article/pii/S2405844023047400]
- 46. Comparative evaluation of ultrasound-assisted extraction ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11904522/]
- 47. Advances in Atmospheric Cold Plasma Technology for ... [https://www.mdpi.com/2304-8158/14/17/2999]
- 48. A comprehensive review of ultrasonic assisted extraction ... [https://www.sciencedirect.com/science/article/pii/S1350417723003589]
- 49. Effect of cold atmospheric plasma pre-treatment and ... [https://www.sciencedirect.com/science/article/pii/S0023643825001355]
- 50. The combined effect of cold atmospheric plasma and ... [https://www.sciencedirect.com/science/article/pii/S002364382500578X]
- 51. Optimization of ultrasonic Bath and cold plasma pre ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10084965/]
- 52. RSM-Based Optimization Analysis for Cold Plasma and ... [https://www.mdpi.com/2304-8158/13/19/3084]
- 53. Comparison of the Conventional, Chemical, and ... [https://www.mdpi.com/2304-8158/14/14/2428]
- 54. The Application of Ultraviolet Treatment to Prolong ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10297300/]
- 55. Current Status of Pulsed UV Light Technology for Food ... [https://link.springer.com/article/10.1007/s12393-025-09428-3]
- 56. Applications of Pulsed Light Decontamination Technology ... [https://www.mdpi.com/2076-3417/10/10/3606]
- 57. Pulsed light sterilization of packaging materials [https://www.sciencedirect.com/science/article/abs/pii/S2214289415000472]
- 58. Hoenle UV Surface Disinfection & Purification Systems [https://www.hoenle.com/products-solutions/applications/ultraviolet-disinfection/surface-disinfection/]
- 59. Impact on Characteristics, Efficacy, and Safety of Foods [https://pmc.ncbi.nlm.nih.gov/articles/PMC10649655/]
- 60. Innovative Non-Thermal Processing Technologies for Shelf ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12427722/]
- 61. Carbon footprint and cost analysis of non-thermal food ... [https://www.frontiersin.org/journals/sustainable-food-systems/articles/10.3389/fsufs.2025.1585467/full]
- 62. Comparative Analysis of Cost, Energy Efficiency, and ... [https://www.mdpi.com/2304-8158/14/13/2239]
- 63. Thermal and Non-Thermal Treatments to Preserve and ... [https://www.mdpi.com/2304-8158/11/21/3400]
- 64. Innovative Non-Thermal Processing Technologies for Shelf ... [https://www.mdpi.com/2304-8158/14/17/2953]
- 65. Thermal Processing - an overview [https://www.sciencedirect.com/topics/engineering/thermal-processing]
- 66. Application of High-Pressure-Based Technologies in the Food ... [https://ohioline.osu.edu/factsheet/fst-fabe-1001]
- 67. The impact of thermal and non-thermal processing on ... [https://www.sciencedirect.com/science/article/pii/S2772502225000423]
- 68. Non-Thermal Technologies in Food Processing [https://www.mdpi.com/2076-3417/15/6/3049]
- 69. Effects of non-thermal processing techniques on cereal bran [https://pmc.ncbi.nlm.nih.gov/articles/PMC12355590/]
- 70. Evaluating the efficacy of non-thermal microbial load reduction ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0283287]
- 71. Advances in non-thermal food processing [https://pubs.rsc.org/en/content/articlelanding/2025/fb/d5fb00136f]
- 72. Nonthermal food processing: A step towards a circular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9684704/]
- 73. Non-Thermal Processing → Term [https://energy.sustainability-directory.com/term/non-thermal-processing/]
- 74. High–Pressure Processing vs. Thermal Treatment: Effect ... [https://www.mdpi.com/2304-8158/10/12/2919]
- 75. Impact of High-Pressure Processing on Antioxidant Activity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8434123/]
- 76. Stability of phenolic compounds in apple and strawberry [https://www.sciencedirect.com/science/article/pii/S0308814622020611]
- 77. TT and HPP in Strawberry Products | Encyclopedia MDPI [https://encyclopedia.pub/entry/17824]
- 78. High pressure processing and thermal pasteurization of strawberry ... [https://link.springer.com/article/10.1007/s13197-017-2529-4]
- 79. Impact of high pressure processing on total antioxidant ... [https://www.sciencedirect.com/science/article/abs/pii/S1466856408001288]
- 80. Control of Enzymatic Browning in Strawberry, Apple, and ... [https://www.mdpi.com/2304-8158/11/13/1942]
- 81. The Effects of High-Pressure Processing Pre-Treatment on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11275511/]
- 82. Effects of Thermal and Non-Thermal Sterilization Methods ... [https://www.mdpi.com/2311-5637/11/2/65]
- 83. The Effects of Thermal and Non - thermal on Vitamin... Processing [https://www.academia.edu/24966540/The_Effects_of_Thermal_and_Non_thermal_Processing_on_Vitamin_C_Carotenoids_Phenolic_Compounds_and_Total_Antioxidant_Capacity_in_Orange_Juice]
- 84. Comparative Analysis of Vitamin, Mineral Content, and ... [https://www.mdpi.com/2223-7747/13/7/1037]
- 85. Evaluation of bioaccessibility and bioavailability of dietary ... [https://www.sciencedirect.com/science/article/abs/pii/S2212429224018595]
- 86. Impact of Thermal Processing on the Protein Digestibility ... [https://www.mdpi.com/2304-8158/14/12/2096]
- 87. Effect of thermal and non-thermal processing on ... [https://www.sciencedirect.com/science/article/pii/S2590157524007843]
- 88. comparative metabolomic analysis of dried loquat ... [https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2025.1637247/full]
- 89. Sensory shelf-life estimation: A review of current ... [https://www.sciencedirect.com/science/article/abs/pii/S0963996912002517]
- 90. Frontiers | Non - thermal Technologies for Food Processing [https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2021.657090/full]
- 91. Non-thermal techniques for microbiological safety, nutritional ... [https://www.ffhdj.com/index.php/FunctionalFoodScience/article/view/1326]
- 92. Sensory Shelf-Life: Key to Quality Control [https://sensesbit.com/blog/sensory-shelf-life-consumer]
- 93. Sensory Shelf-Life Test [https://www.sensorysociety.org/knowledge/sspwiki/Pages/Sensory%20Shelf-Life%20Test.aspx]
- 94. A Sensory Shelf-Life Study for the Evaluation of New Eco ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11083942/]
- 95. Principles and recent applications of novel non - thermal ... processing [https://pubmed.ncbi.nlm.nih.gov/30580554/]